Invented by David M. Goldenberg, Serengulam V. Govindan, Immunomedics Inc
The use of ADCs in cancer therapy has gained significant attention in recent years due to their ability to address some of the limitations of traditional chemotherapy. Unlike conventional chemotherapy drugs that often have a broad spectrum of activity, ADCs are designed to specifically target cancer cells that express a particular antigen. This targeted approach not only increases the efficacy of the treatment but also reduces the risk of side effects associated with non-specific drug distribution.
The market for ADCs is expected to witness substantial growth in the coming years. According to a report by Grand View Research, the global ADC market size was valued at $2.9 billion in 2020 and is projected to reach $13.4 billion by 2028, growing at a compound annual growth rate (CAGR) of 20.7%. This growth can be attributed to several factors, including the increasing prevalence of cancer, advancements in antibody engineering, and the growing number of clinical trials evaluating ADCs.
ADCs have shown promising results in the treatment of various types of cancer, including breast cancer, lymphoma, and lung cancer. One of the most well-known ADCs is Trastuzumab emtansine (T-DM1), which is approved for the treatment of HER2-positive breast cancer. T-DM1 combines the anti-HER2 antibody trastuzumab with the cytotoxic drug emtansine, resulting in improved efficacy and reduced toxicity compared to traditional chemotherapy regimens.
In addition to their use as standalone therapies, ADCs are also being explored in combination with other treatment modalities, such as immune checkpoint inhibitors and radiotherapy. The combination of ADCs with immunotherapies has the potential to enhance the anti-tumor immune response and improve patient outcomes.
Despite the promising potential of ADCs, there are still challenges that need to be addressed. One of the main challenges is the development of effective linkers that can stably attach the antibody and the drug payload, ensuring optimal drug release at the tumor site. Additionally, the manufacturing process of ADCs is complex and requires specialized expertise, which can limit their widespread adoption.
In conclusion, the market for Antibody-drug conjugates is rapidly expanding, driven by their potential to revolutionize cancer treatment. ADCs offer a targeted approach to delivering potent drugs directly to cancer cells, minimizing damage to healthy tissues. With ongoing advancements in antibody engineering and increasing clinical trials, ADCs are expected to play a significant role in the future of cancer therapy. However, further research and development are needed to overcome challenges and ensure the widespread availability of these innovative treatments.
The Immunomedics Inc invention works as follows
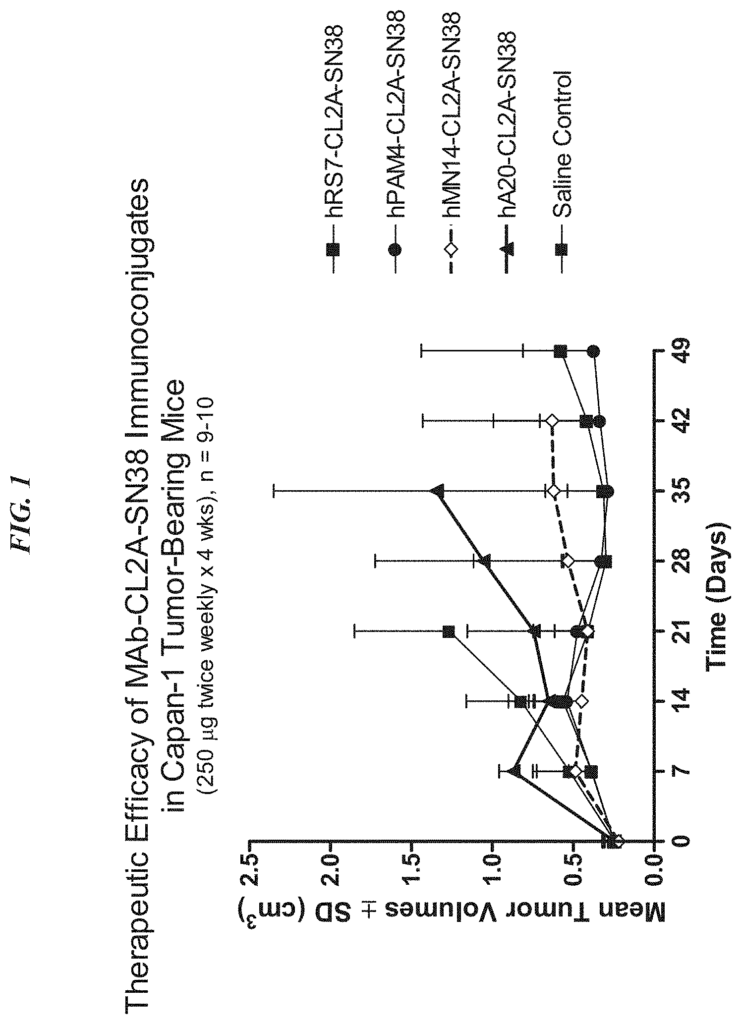
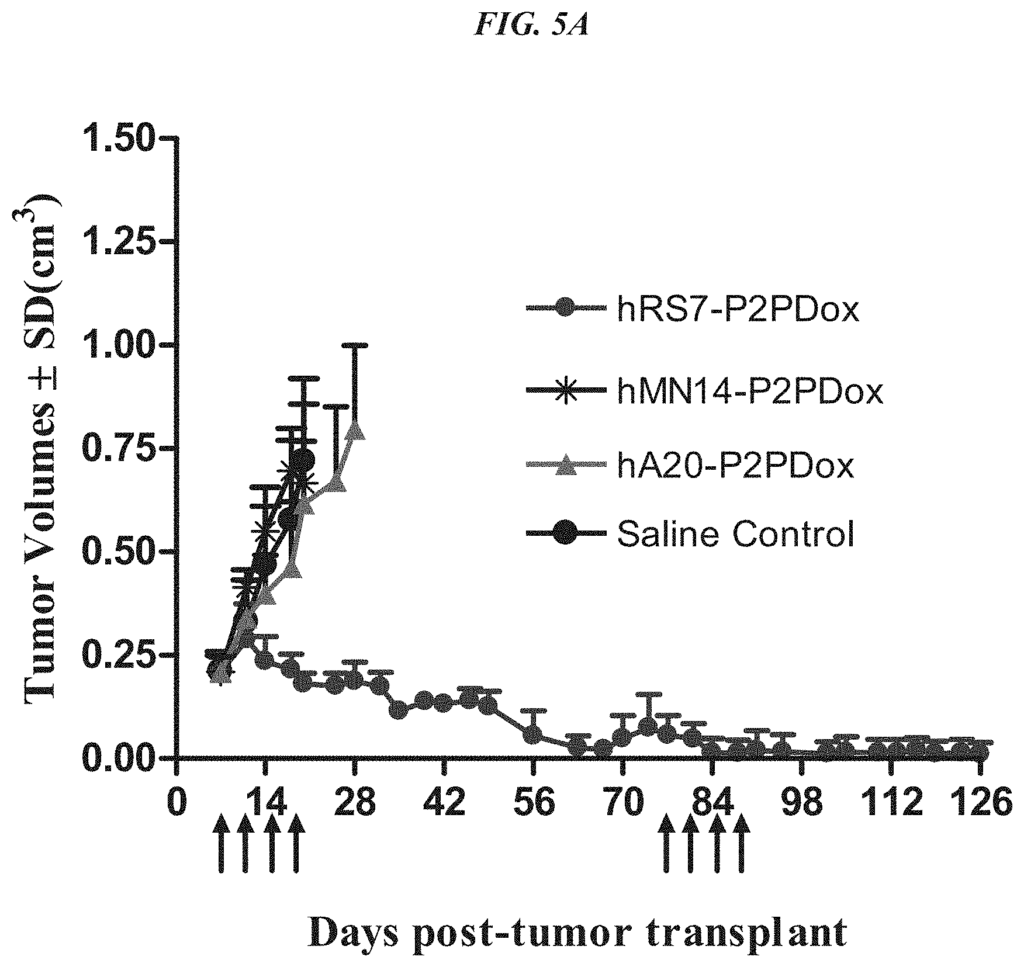
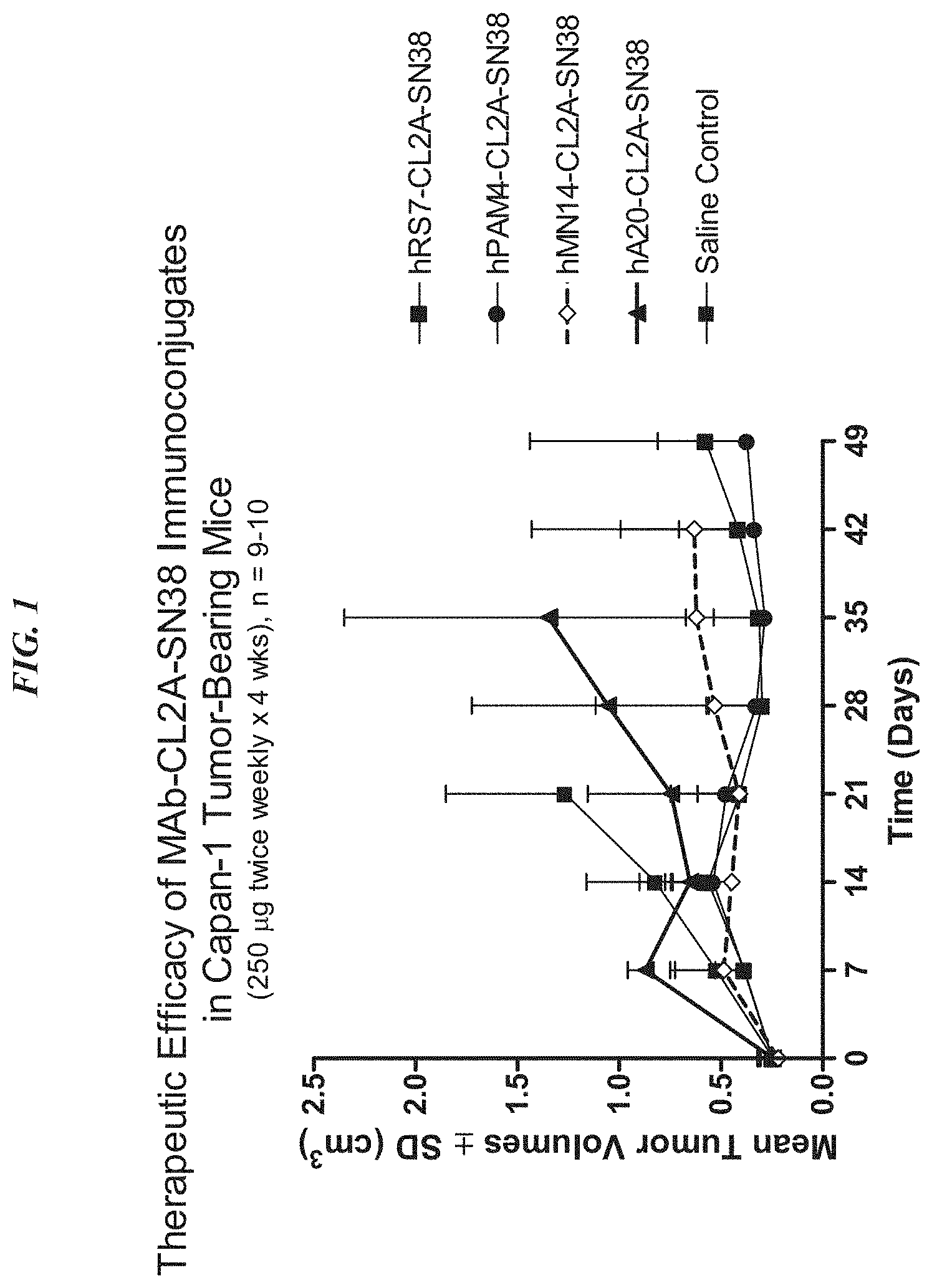
The present invention relates therapeutic immunoconjugates containing SN-38 coupled to an antigen-binding fragment or antibody. The immunoconjugate can be given at doses between 4 mg/kg to 16 mg/kg. The immunoconjugate, when administered according to a schedule and dosage, can reduce the size of solid tumors, reduce or eliminate metastatic growth, and treat cancers that are resistant to standard treatments, such as chemotherapy, radiation therapy or immunotherapy. The immunoconjugate can be used to treat cancers which are resistant to irinotecan or have relapsed after treatment.
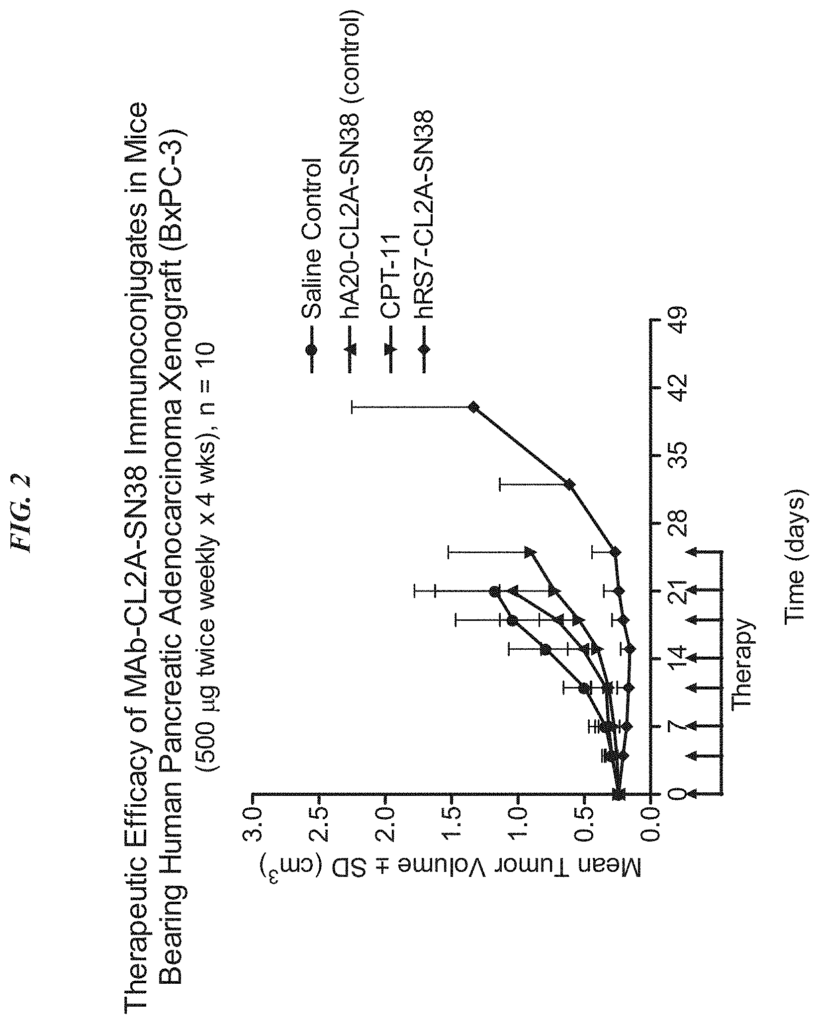
Background for Antibody-drug conjugates, and their uses
The present invention relates to the treatment of cancer using antibody-drug conjugates in various embodiments. ADCs can be used as an alternative or in combination with other therapeutic methods, including surgery, radiation, chemotherapy, immunomodulators and cytokines. Other therapies include drugs, toxins or radionuclides. ADC is used in preferred embodiments to treat cancers that are resistant to standard treatments, such as metastatic breast cancer, metastatic colorectal carcinoma, or metastatic pancreatic tumors. The combination of ADC with other therapeutic modality has a greater efficacy than either one alone or the summation of individual treatment effects.
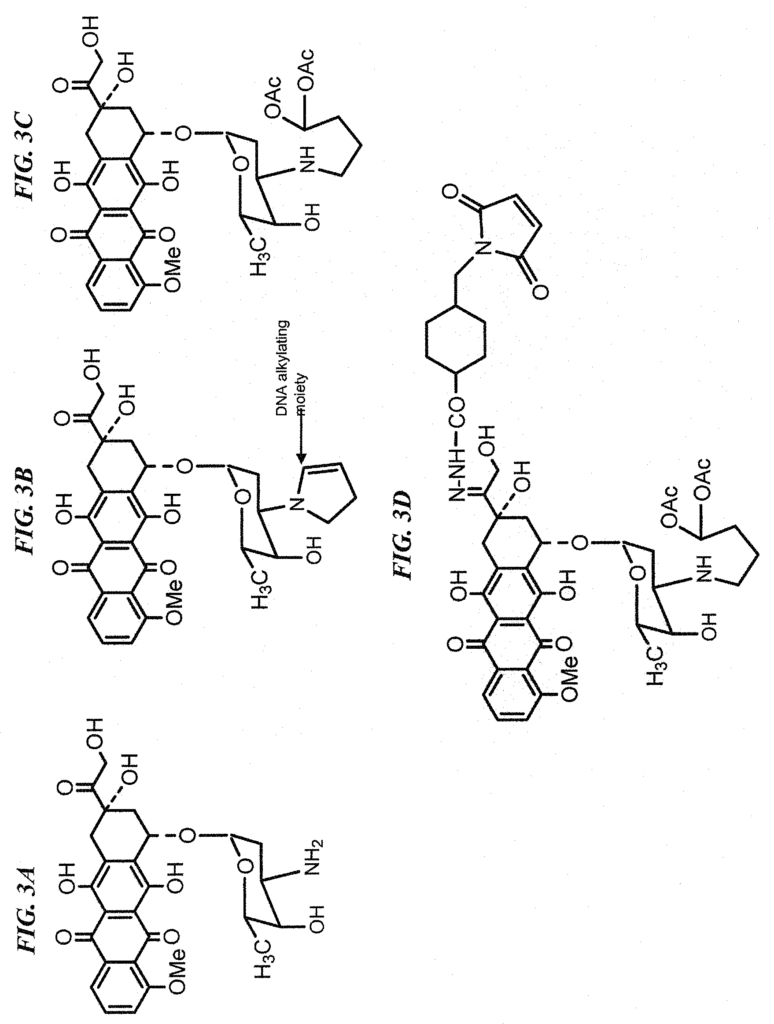
In a particular embodiment, an antiTrop-2 antigen may be a humanized RS7 antigen (see, e.g. U.S. Pat. No. No. As discussed below, other anti-Trop-2 antibody may be used to make the ADCs. Many cytotoxic cancer drugs are known in the art, and any of these drugs can be conjugated with the antibody. In a more preferred embodiment, the drug conjugated to the antibody is a camptothecin or anthracycline, most preferably SN-38 or a pro-drug form of 2-pyrrolinodoxorubicin (2-PDox) (see, e.g., U.S. patent application Ser. Nos. Nos.
In another preferred aspect, therapeutic conjugates comprising anti-CEACAM5 antibodies (e.g. hMN-14 or labretuzumab), and/or anti-CEACAM6 antibodies (e.g. hMN-3, hMN-15), may be used to treatment any of a number of cancers expressing CEACAM5 and/or CEACAM6, according to U.S. Nos. The Examples sections of 7,541,440, 7,951,369, 5,874,540, 6,676,924 et 8,267.865 are incorporated by reference. Solid tumors which may be treated with anti-CEACAM5, anti CEACAM6, or both include, but are not restricted to, breast, lung and pancreatic cancers, as well as ovarian, colon and rectum cancers, bladder, mouth, stomach, and urinary bladder. The subject immunoconjugates can be used to treat a majority of carcinomas including gastrointestinal and respiratory cancers, as well as genitourinary, breast and ovarian cancers. The hMN-14 is a humanized antigen that contains the light chain variable CDR sequences (KASQDVGTSVA, SEQID NO:114), heavy chain variable CDR (TYWMS, SEQID NO:99), and CDR3 sequences (LYFGFPWFAY, SEQID NO:101). The hMN-3 is a humanized antigen that consists of light chain variable region sequences (CDR1:RSSQSIVHSNGNTYLE; SEQ NO:102), (KVSNRFS: SEQ NO:103), (FQGSHVPPT: SEQ NO:104), and (KVSNRFS: SEQ NO:103), and heavy chain sequences (CDR1:NYGMN; SEQ NO:105); C The hMN-15 is a humanized antigen that contains light chain variable regions CDRs SASSRVSYIH, GTSTLAS, and QQWSYNPPT. It also includes heavy chain variable regions CDRs DYYMS, FIANKANGHTTDYSPSVKG and DMGIRWNFDV.
The antibody moiety can be a monoclonal, bispecific, multivalent, or any other antibody-based molecules. The antibody may be of different isotypes. Preferably, it will contain human IgG1, IgG2, IgG3, or IgG4 hinge and constant regions. The antibody or fragment can be humanized or chimeric as well as variations such as half IgG4 antibodies. ), as described by van der Neut Kolfschoten et al. (Science 2007; 317:1554-1557). The antibody or fragment thereof can be selected or designed to contain human constant region sequences belonging to specific allotypes. This may reduce immunogenicity in the ADC when administered to human subjects. Allotypes that are preferred for administration include non-G1m1 (nG1m1) allotypes, such as G1m3, G1m3,1, G1m3,2, or G1m3,1,2. The allotype should be selected from the nG1m1, nG1m1,2, and Km3 groups.
The drug that will be conjugated with the antibody or fragment of the antibody can be chosen from the following: anthracycline (or camptothecin), a tubulin antagonist, calicheamycin (or maytansinoid), a triazene (or nitrosourea), a taxane analog, COX-2 inhibitors, epipodophyllotoxins, platinum coordination complexes, vinca alkaloids, substituted ureas
Specific drugs of use may be selected from the group consisting of 5-fluorouracil, afatinib, aplidin, azaribine, anastrozole, anthracyclines, axitinib, AVL-101, AVL-291, bendamustine, bleomycin, bortezomib, bosutinib, bryostatin-1, busulfan, calicheamycin, camptothecin, carboplatin, 10-hydroxycamptothecin, carmustine, celecoxib, chlorambucil, cisplatinum, COX-2 inhibitors, irinotecan (CPT-11), SN-38, carboplatin, cladribine, camptothecans, crizotinib, cyclophosphamide, cytarabine, dacarbazine, dasatinib, dinaciclib, docetaxel, dactinomycin, daunorubicin, DM1, DM3, DM4, doxorubicin, 2-pyrrolinodoxorubicine (2-PDox), a pro-drug form of 2-PDox (pro-2-PDox), cyano-morpholino doxorubicin, doxorubicin glucuronide, endostatin, epirubicin glucuronide, erlotinib, estramustine, epidophyllotoxin, erlotinib, entinostat, estrogen receptor binding agents, etoposide (VP16), etoposide glucuronide, etoposide phosphate, exemestane, fingolimod, floxuridine (FUdR), 3?,5?-O-dioleoyl-FudR (FUdR-dO), fludarabine, flutamide, farnesyl-protein transferase inhibitors, flavopiridol, fostamatinib, ganetespib, GDC-0834, GS-1101, gefitinib, gemcitabine, hydroxyurea, ibrutinib, idarubicin, idelalisib, ifosfamide, imatinib, lapatinib, lenolidamide, leucovorin, LFM-A13, lomustine, mechlorethamine, melphalan, mercaptopurine, 6-mercaptopurine, methotrexate, mitoxantrone, mithramycin, mitomycin, mitotane, monomethylauristatin F (MMAF), monomethylauristatin D (MMAD), monomethylauristatin E (MMAE), navelbine, neratinib, nilotinib, nitrosurea, olaparib, plicomycin, procarbazine, paclitaxel, PCI-32765, pentostatin, PSI-341, raloxifene, semustine, SN-38, sorafenib, streptozocin, SU11248, sunitinib, tamoxifen, temazolomide, transplatinum, thalidomide, thioguanine, thiotepa, teniposide, topotecan, uracil mustard, vatalanib, vinorelbine, vinblastine, vincristine, vinca alkaloids and ZD1839.
The optimal dosing schedule may include two consecutive weeks of therapy followed by one, two, three or four weeks of rest, or alternate weeks of therapy and rest. The optimal schedule of dosing may include two consecutive weeks’ therapy, followed by alternating weeks between therapy and rest. It could also include a week of treatment followed by two or three or even four weeks rest. Or, it could be a week of treatment followed by alternating weeks rest or therapy. The treatment can be extended to any number of cycles. Preferably, at least 2, but preferably at most 4, or at the very least 6, or at least 8, or at least 10, and at least 12 cycles. Examples of dosages include 1 mg/kg or 2 mg/kg. Other examples are 3 mg/kg or 4 mg/kg. The preferred dosages are 4, 8, 9, 10, 14, 16, or 18 mg/kg. The preferred dosages include 6-12, 6-8 mg/kg, 8-10 mg/kg, or 8-12 mg/kg. A person with ordinary skill would realize that many factors such as age and general health as well as specific organ function and weight as well as the effects of previous therapy on specific organs (e.g. bone marrow), may be taken into consideration when selecting the optimal dosage of ADC. The dosage and/or the frequency of administration can be changed during therapy. The dosage can be repeated as necessary, and tumor shrinkage is evident after only 4-8 doses. The optimized doses and schedules disclosed herein have shown unexpectedly superior efficacy in humans and reduced toxicity, which was not predicted by animal model studies. The superior efficacy allows for the treatment of tumors previously resistant to standard anti-cancer treatments. The treatment was found to be effective against tumors previously resistant camptothecins such as irinotecan (the parent compound of SN-38)”.
The ADCs can be used to treat cancers such as breast cancer, ovarian cancer, cervical cancer, endometrial cancer, prostate cancer, colon cancer, esophageal cancer, bladder cancer, thyroid cancer, epithelial cancer, or head and neck cancer. The ADC is particularly useful for cancers resistant to standard anti-cancer treatments, such as metastatic colon, triple-negative, breast, prostate, lung, kidney, pancreatic, thyroid, epithelial, head-and-neck, and ovarian cancers.
Definitions
If not otherwise specified, the?an? or?a? “Unless otherwise specified,?a? “One or more
As used herein, ?about? means plus or minus 10%. “About 100” is a good example. “Any number between 90 and110” would be included in the phrase “about 100”.
An antigen, as described in this document, is a full-length immunoglobulin (i.e. naturally occurring or created by normal immunoglobulin fragment recombinatorial process) molecule (e.g. an IgG) or a portion immunologically active, i.e. specifically binding, of an immunoglobulin (like an antibody fragment).
An antigen fragment is a part of an antibody, such as F(ab?) “An antibody fragment is a portion of an antibody such as F(ab? As discussed below, antibody fragments can also include IgG4 halves and single domain antibodies. An antibody fragment, regardless of its structure, binds to the same antigen as the full-length antibodies. The term “antibody fragment” is used. The term ‘antibody fragment’ also refers to isolated fragments consisting only of the variable regions in antibodies, like the?Fv” The variable regions of both the heavy and the light chains are fragmented, as well as recombinant monopeptide molecules containing the light and heavy variable region connected by a linker peptide (?scFv protein ?).”).
The constant domains are derived by recombinant proteins from human antibodies. “A chimeric antigen is a recombinant molecule that contains the variable regions, including the complementarity-determining regions (CDRs), of an antibody derived preferably from a rodent. The constant domains can be taken from another species such as a dog or cat for veterinary purposes.
Humanized antibodies are recombinant proteins in which CDRs of an antibody from a species, such as a rodent, have been transferred from heavy and lighter variable chains into heavy and lighter variable domains of human (e.g. framework region sequences). The constant domains are derived by a human antibody. In certain embodiments a limited number framework region amino acids from the rodent antibody can be substituted in the human antibody frame region sequences.
A human antigen is, for example, an antibody produced by transgenic mice which have been “engineered” To produce specific human antibody in response to an antigenic challenge. This technique involves the introduction of human heavy- and light-chain loci into mouse strains derived from embryonic cell lines with targeted disruptions to the murine heavy- and lightchain loci. Transgenic mice are able to synthesize human antigen-specific antibodies, and they can also be used to create hybridomas that secrete human antibodies. Green et. al. describe methods for obtaining human antibody from transgenic mouse. 7:13 (1994), Lonberg et al., Nature 368:856 (1994), and Taylor et al., Int. Immun. 6:579 (1994). The art also allows for the construction of a fully human antibody using genetic or chromosomal methods as well as phage displays. McCafferty, et. al., Nature, 348:552-553, (1990), for example, describes the in vitro production of human antibody fragments and antibodies from immunoglobulin gene repertoires from non-immunized donors. This technique involves cloning antibody variable domains in-frame to either a major coat protein or minor coat gene of a filamentous bacteria phage, resulting in functional antibodies fragments being displayed on the surface. The filamentous particle contains the single-stranded copy of the phage’s genome. Selections made based on functional properties of an antibody will also select the gene that encodes the antibody with those properties. The phage can mimic some of the characteristics of the B-cell in this way. For a review of the different formats in which phage display can be done, please see, e.g. Johnson and Chiswell Current Opinion in Structural Biology, 3:5564-571 ( 1993). In vitro activated cells of B can also produce human antibodies. U.S. Pat. Nos. Nos.
A therapeutic agent can be a compound or atom that is used to treat a particular disease. It may be administered in conjunction with or separately from an antibody moiety. Therapeutic agents include antibodies, fragments of antibodies, drugs, nucleases and hormones. They can also be immunomodulators, proapoptotic agents or anti-angiogenic, boron compounds. Below, we describe the therapeutic agents in greater detail.
An immunoconjugate can be an antibody, an antibody fragment, or a fusion protein that is conjugated with at least one diagnostic and/or therapeutic agent.
A multispecific antibody can bind to two or more targets of different structures, such as two antigens or two epitopes. It may also bind to haptens and/or antigens or epitopes. Multivalent, multispecific antibodies are constructs with more than one specific binding site.
A bispecific antigen is an antibody which can bind to two targets simultaneously. Bispecific antibodies and bispecific fragments of antibody (bsFabs) can have one arm that binds specifically to, for instance, a tumor associated antigen, and another arm that binds specifically to a targetable fusion protein that contains a diagnostic or therapeutic agent. A variety of bispecific proteins can be created using molecular engineered.
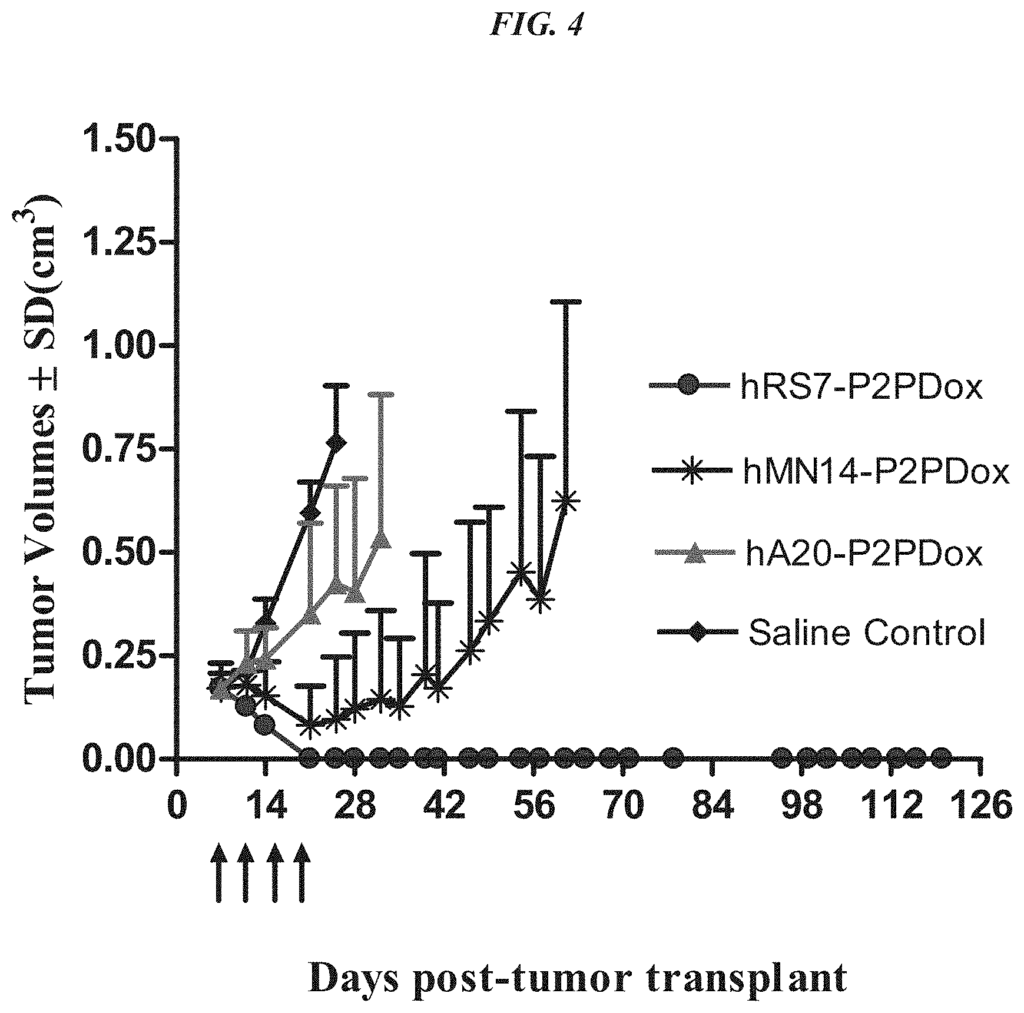
Anti-Trop-2 Antibodies
Click here to view the patent on Google Patents.
Leave a Reply