Invented by Andrew David Griffiths, David A. Weitz, Darren Roy Link, Keunho Ahn, Jerome Bibette, United Kingdom Research and Innovation, Harvard College
The United Kingdom Research and Innovation, Harvard College invention works as follows
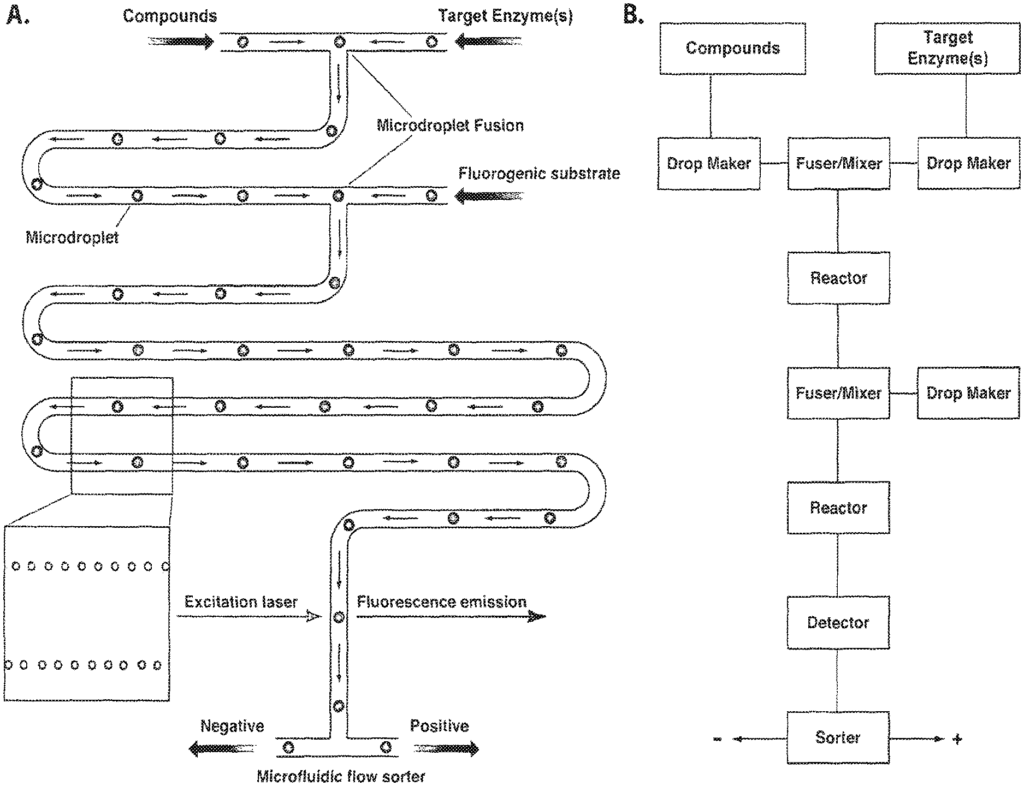
The invention is a method of identifying compounds that bind to the target component of a biological system or modulate its activity. It comprises the steps: a), compartmentalizing the compounds in microcapsules with the target so that only a small subset is represented multiple times in each microcapsule, and b), identifying the compound that binds to the target or modulates its activity; wherein, at least one of these steps is performed under control microfluidic. The invention allows the screening of large libraries of molecules that can be used as drug leads.
Background for Compartmentalised Screening by Microfluidic Control
The present invention is a method for identifying molecules that bind to the target component in a biochemical system, or modulate its activity.
Over the last decade, high-throughput (HTS), screening of compound libraries became a cornerstone of pharmacological research. HTS investment is significant. According to a current estimate, biological screening and preclinical testing account for about 14% of total R&D expenditures in the pharmaceutical industry. (Handen Summer 2002). HTS has improved significantly in recent years due to the need to reduce costs and increase screening targets and compounds. The 96-well format has been largely replaced by formats such as 384-wells, 1536-wells and even 3456 wells. Combining this with plate-handling robots, which are commercially available, allows for the screening of 100,000 or more assays each day. This reduces costs per test due to miniaturization.
HTS is supported by other innovations. Combinatorial chemistry can be used to create large numbers of structurally similar compounds for HTS. At the moment, most combinatorial synthesis involves spatially-resolved parallel synthesis. The number of compounds which can be synthesized is limited, but they can be synthesized on a milligram or tens-of-milligram scale allowing for full characterisation and purification. Split synthesis can be used to create larger libraries by generating one-bead, one compound libraries. This method is less popular due to several limitations, including the need for solid-phase synthesis, the difficulty of characterizing the final products because of the sheer numbers and the small scale; the small amount of compound on each bead only being sufficient for a single or few assays; and the difficulty of identifying the structure for a hit compound which relies often on tagging and encoding methods that complicate both synthesis or analysis. Split synthesis and single-bead analysis are still promising despite this. Recent developments have seen significant improvements in single bead and miniaturised screen analysis. Printing techniques, for example, allow binding assays on a slide with 10,800 spots of compound, each 1 nl in volume (Hergenrother and al., 2000). Combichem, to date, has only produced a small number of lead compounds. In April 2000, only ten compounds with a history of combinatorial chemical synthesis had been developed into clinical trials. All but three are (oligo-)nucleotides and peptides. In fact, despite huge investments in HTS and Combinatorial Chemistry during the last decade, the number of new drug introductions per year has been at best constant.
The size of the libraries that have been created and screened so far (40,000) is still relatively small.
Virtual screening (VS) is a computational approach that searches large compounds bases to identify subsets of candidates molecules. This can be useful in conjunction with HTS. There are few studies to date that compare the performance of HTS and VS. Further validation is needed.
Microfluidic technologies has been applied to high-throughput screening methods.” U.S. Pat. No. No. U.S. Pat. No. No.
Despite these advances, the current screening throughput still isn’t adequate. According to recent estimates, there are 30,000 genes in the genome of humans. This combined with the thousands of chemical structures that can be created using existing chemistry suggests that a large number of tests would be needed to map the space of structure-activity for all possible therapeutic targets.
Therefore, a screening method that allows for the rapid screening of large numbers (?109), or smaller numbers, of compounds in a variety of conditions (different compounds concentrations, targets, etc.), would be very useful. The ability to generate novel drug leads using only reaction volumes as small as a few femtolitres at a very low cost is a huge advantage.
Tawfik and Griffiths (2000), and International Patent Application PCT/GB98/01889 describe a system of in vitro evolution that uses compartmentalisation within microcapsules in order to link genotype and the phenotype on a molecular scale. In Tawfik & Griffiths (1998) and in various embodiments of International Patent Application PCT/GB98/01889 the desired activity of gene products results in a change in the genetic element that encoded it. In a later step, the modified genetic element is selected.
The present invention also relies on compartmentalisation within microcapsules. In this case, compartmentalisation is based upon compounds selected from a library of compounds. The microcapules consist of droplets of liquid that are made and manipulated by using systems and techniques for controlling fluidic species, and in particular by systems and procedures for electronic control of fluidic substances.
The manipulation fluids in order to create fluid streams with desired configurations, discontinuous streams, droplets or particles, dispersions etc. for fluid delivery, product manufacturing, analysis and other purposes is a relatively studied art. Capillary flow focussing, for example, has been used to produce highly monodisperse bubbles of less than 100 microns diameter. This technique involves forcing gas out of a tube of liquid into a bath. The tube is then positioned over a small hole, and the contraction of the liquid outside the orifice concentrates the gas into a thin, jet-like jet that breaks into bubbles of equal size via capillary instability. A similar arrangement was used in a related technique to produce liquid droplets.
An article entitled ‘Generation Of Steady Liquid Microthreads And Micron-Sized Monodisperse sprays and Gas Streams? Phys. Rev. Lett. 80:2, January 12, 1998, 285-288. (Ganan Calvo) describes the formation of a microscopic thread of liquid by a laminar gas stream accelerating. This gives rise to a fine mist.
An article entitled,?Dynamic pattern formation in a vesicle-generating microfluidic device? Phys. Rev. Lett., 86:18, Apr. 30, 2001 (Thorsen, et al.) The formation of a discontinuous oil phase by microfluidic crossingflow is described in Thorsen, et al., 2001. The formation of a discontinuous water phase in a continuous oil phase via microfluidic crossflow is described. Specifically, by introducing water at?T?
U.S. Pat. No. No. 6,120,666, published Sep. 19,2000, describes a microfabricated device with a fluid-focusing chamber for spatially confining the first and second fluid samples for analysing microscopic particles within a liquid medium, such as in biological fluid analysis.
U.S. Pat. No. No.
U.S. Pat. No. No.
U.S. Pat. No. No. “19, 2001 describes the production of particles to be introduced into food by using a microjet. A monodisperse aerosol is formed when the microjet detaches.
Microfluidic Systems have been described in various contexts. Typically, they are used in miniaturized laboratory analysis (e.g. clinical). There have also been other uses described. International Patent Publication No. The International Patent Publication No. WO 01/89789 by Anderson et. al. published on Nov. 29, 2001, describes a multi-level microfluidic system that can be utilized to create patterns of materials such as biological materials or cells, onto surfaces. “Other publications describe microfluidic system components such as valves, switches and other components.
While significant progress has been made on the macro- or microfluidic level, better techniques and results from these techniques are required.
We have developed a method for screening compounds that are not encoded with genetic elements using a compartmentalised system based on the one described by Griffiths & Tawfik (2000). The present invention uses fluidic species control systems and methods to allow rapid, high-throughput testing of compounds against a target. This is done at low cost using modern HTS techniques.
These systems and methods are extremely useful for screening compounds, as they: