Invented by Michael M. Lipp, Jean C. Sung, Pulmatrix Operating Co Inc
The Pulmatrix Operating Co Inc invention works as follows
The present invention is a method of treating a patient using respirable powders. These powders contain dry particles that are composed of about 20%-37.5 % (w/w), leucine and 58.6-75% (w/w), calcium lactate.
Background for Respirable dry powder containing calcium lactate, sodium Chloride and Leucine
Pulmonary administration of therapeutic agents has several advantages over alternative delivery methods. The advantages of inhalation include the rapid onset of action, convenience for the patient, potential reduced side effects, ease of use, elimination of needles and more. Inhalation therapy can be used in a hospital or clinic setting. It is quick and easy to administer, has minimal side effects, and is very convenient.
The respiratory tract is delivered therapeutic agents using “Metered Dose Inhalers” (MDIs). MDIs can be used to administer therapeutic agents that are formulated in the form of solid particles or dry powders suspended in a volatile fluid under pressure. The suspension is released at a relatively high speed by opening a valve. The liquid volatilizes and leaves behind a fast moving aerosol of dry particle that contains the therapeutic agent. MDIs deliver drugs to upper and middle airways, but they are limited by the fact that they only deliver small doses of drug per actuation. The bronchioles, and the alveoli are the most common sites of pulmonary disease manifestations such as infections and asthma.
Liquid Aerosol Delivery is one of oldest forms of pulmonary drugs delivery. Air jet nebulizers typically create liquid aerosols by releasing compressed air at high speed through a small orifice. This results in low pressure near the exit due to the Bernoulli Effect. See, e.g., U.S. Pat. No. 5,511,726. Low pressure is used for drawing the fluid out of a tube. As it moves through the air, this fluid is broken down into smaller droplets. This standard nebulizer’s disadvantages include: (i) a relatively large liquid aerosol primary droplet, which often requires impaction onto a baffle in order to produce secondary splash droplets that are respirable, (ii), a lack of uniformity in liquid aerosol size, (iii), significant recirculation in bulk drug solution and (iv), low densities in inhaled air of small, respirable liquid aerosol drops.
Ultrasonic Nebulizers” use piezoelectric disks that are flat or concave and submerged beneath a liquid reservoir. They resonate the surface of this liquid reservoir to form a liquid cone, which then sheds aerosols from its surface. (U.S. No. 5,551,416). Aerosolization does not require airflow, so high concentrations of aerosol can be achieved. The piezoelectric components, however, are expensive to manufacture and inefficient in aerosolizing suspensions. This means that the active drug must be dissolved in low concentrations of water or saline solution. The newer liquid aerosol technology involves generating smaller, more uniform, and respirable liquid dry particles through passing the liquid that is to be aerosolized though micron-sized hole. See, e.g., U.S. Pat. No. 6,131,570; U.S. Pat. No. No. 5,724,957, and U.S. Patent. No. 6,098,620. This approach has several disadvantages, including the relatively expensive fine mesh and piezoelectric components and fouling of holes by residual salts or solid suspensions.
Dry powder inhalation relies on the lactose blend to ensure that particles are dispersed enough to inhale, but not small enough for inhalation. It is well-known that this process is inefficient and does not work with some drugs. Many groups have attempted to overcome these limitations by developing dry powder formulations (DPIs) that are dispersible, respirable, and do not require the lactose blend. U.S. Patent describes dry powder formulations used for inhalation therapies. No. No. 5,993,805 Sutton et al. ; U.S. Pat. No. 6,9216527 to Platz et al. WO 0000176 was issued to Robinson et al. ; WO 9916419 to Tarara et al. ; WO 0000215 to Bot et al; U.S. Pat. No. 5,855,913 to Hanes et al. U.S. Pat. Nos. Nos.
The wide clinical application of dry-powder inhalation has been restricted by the difficulties of generating powders with the appropriate particle size, density and dispersibility. It is also difficult to keep the powder dry and develop a hand-held device which can effectively disperse the respirable, dry particles that are inhaled into the air. The particle size of the dry powders used for inhalation is also limited because smaller respirable particles are more difficult to disperse into air. While dry powder formulations offer advantages over liquid dosage forms or propellant-driven formulations they are susceptible to aggregation, and have low flowability. This reduces the dispersibility of inhalation therapy using dry powders. Dry particles can aggregate due to interparticular Van der Waals interaction and capillary condensing effects. Hickey A. and colleagues, “Factors Affecting the Dispersion Of Dry Powders As Aerosols”, Pharmaceutical Technology, August 1994. “Maintaining the physicochemical characteristics of dry powder over time and during long-term storage is a challenging task.
To overcome interparticle adhesive forces, Batycky et al. In U.S. Pat. No. No. The volume median geometric size (VMGD), as measured by a laser diffraction system such as the HELOS/RODOS (manufactured in Princeton, N.J.), is greater than 5 microns. Batycky, et. al., column 7 lines 42-65. A second approach to improving the dispersibility for respirable particles with an average particle size less than 10?m involves adding a water-soluble polypeptide, or other excipients suitable (including amino acids excipients like leucine), in an amount ranging from 50% to 99.9% of the total composition. See Eljamal et al., U.S. Pat. No. No. This approach, however, reduces the amount active agent that can delivered with a fixed quantity of powder. To achieve the desired therapeutic effects, it is necessary to use a larger amount of powder, such as multiple inhalations or frequent administration. Other approaches use devices that apply mechanical pressure, such as compressed gasses to the small particle to disrupt interparticular adhesiveness during or just before administration. See, e.g., U.S. Pat. No. 7,601,336 to Lewis et al., U.S. Pat. No. No. 6,737,044 Dickinson et al. No. No. Johnston et al.
Another limitation shared by all of the methods above is that aerosols produced in this way typically contain substantial quantities of non-drug materials such as solvents, emulsifiers and propellants. The large amounts of non-drug materials are needed to form dry particles that are small enough to be delivered into the alveoli (e.g. Less than 5 microns, and preferably below 3 microns. These amounts of nondrug material reduce the amount and purity of active drug substances that can be delivered. These methods are unable to accurately deliver large doses of active drugs for systemic administration.
Therefore there is still a requirement for small particle size aerosols which are highly dispersible. Dry powders dense in both mass and drug are needed to maximize the amount of drug in a container. A method that produces aerosols with greater amounts of drug material and less non-drug materials is also needed. Dry powders with stable physicochemical characteristics are also needed. “Finally, we need a way to allow a patient administer a unit dose quickly with just one or two small volume breaths.
The invention is a dry powder that contains calcium lactate, leucine and sodium chloride. The respirable powders are composed of dry particles that range from about 20% (w/w), to about 37.5%, leucine. They also contain calcium lactate ranging from about 58.6%, to about 75%, and sodium chloride ranging between about 3.9%, to about 5%. A dry powder containing dry particles containing i) approximately 20% (w/w), ii), about 75%(w/w), calcium lactate and iii), about 5% (w/w), sodium chloride, can be an exemplary. A second exemplary dry powder may contain dry particles containing i) approximately 37.5% (w/w), ii), about 58.6%(w/w), calcium lactate and iii), about 3.9% (w/w), sodium chloride. Unless otherwise stated, the compositions of the disclosed formulations are shown on a dried (anhydrous basis).
The invention also relates dry powders which contain dry particles that are composed of calcium lactate, Sodium chloride, an additional therapeutic agent, and optionally Leucine.
The respirable dry particle has a volume-median geometric diameter (VMGD), which is less than 5 microns, as measured by the HELOS/RODOS system at the single bar dispersion settings. The respirable particles have a VMGD less than 5 microns. This could be between 1 and 3 m, measured on the HELOS/RODOS Laser Diffraction System.
The respirable dry powders should have a Hausner ratio of at least 1,5, and preferably 2.0. In certain embodiments, dry powders are at least 1.4 Hausner Ratio. The respirable powders have a ratio of dispersion at 1 bar/4 bars that is less than 1.5. This can be between 1.0 and 1.2. These values were measured using the HELOS/RODOS system’s 1 bar and 4-bar dispersion settings. “The respirable dry powders are less than 1.5 at 0.5 bar/4bar, such as 1.0 to 1.3. This is measured using laser diffraction on the HELOS/RODOS laser diffraction system.
The respirable powders are dry powders with a Fine Particle Fraction of less than 3.4 Microns, which is at least 20%. The respirable powders have a fine particle fraction (FPF), less than 5.6microns, of at least 30 or 40% or 50%.
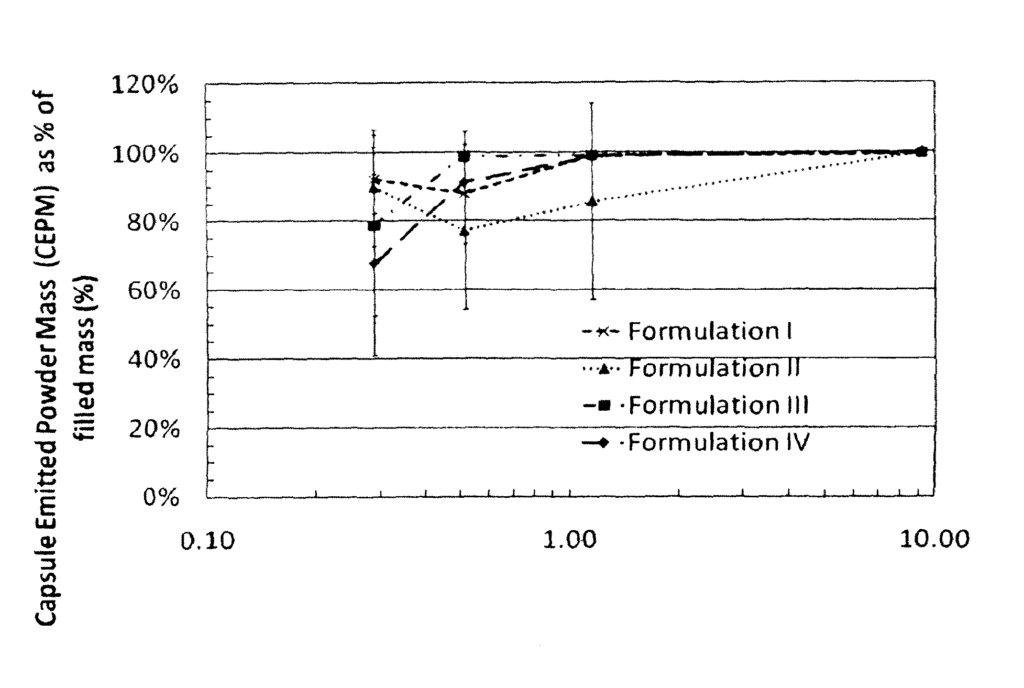
The respirable powders are characterized as having a high dose emitted. A Capsule Emitted Powder Mass of at least about 80 percent of the respirable powder contained in an inhaler that contains 50mg of dry powder is achieved by applying a total energy of inhalation of less than 1 Joule to the dry powder inhaler. A CEPM of about 80% of the respirable dry-powder contained in a container containing 40 mg of dry powder is also possible when applying a total energy of about 1 Joule to a dry-powder inhaler.
The respirable powders may contain crystalline or amorphous states. Calcium lactate, for example, can be either amorphous or crystalline and sodium chloride, leucine, and/or other components can also be amorphous. The sodium chloride and calcium lactate are amorphous, and leucine can be either crystalline or amorphous.
The dry powders that can be inhaled may also contain an additional therapeutic agent.
The invention also relates a method of treating a respiratory disorder, which involves administering an effective amount to a patient’s respiratory tract a respirable powder described herein.
The invention also relates a method of treating or preventing a severe exacerbation in a respiratory condition by administering an effective dose of a dry powder respirable to a patient who is in need of it.
The invention also relates a method of treating or preventing an infection of the respiratory system, which involves administering an effective amount to the respiratory system of a patient who is in need of it.
The invention also relates a method of reducing inflammation, which comprises administering an effective amount to a respiratory tract of a needy patient of a respirable powder described herein.