Invented by Bernard Vallot, Sartorius Stedim FMT SAS
The Sartorius Stedim FMT SAS invention works as follows
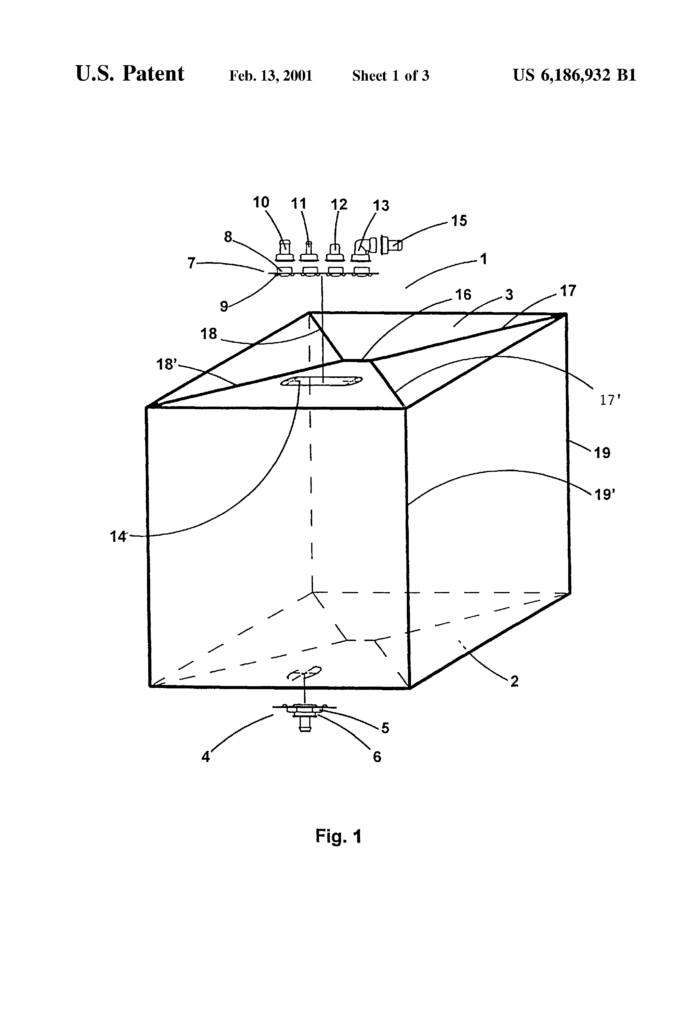
When filled, a flexible sachet of the bellows-type for transporting biopharmaceutical fluids of a volume greater than 50 liters assumes a parallelepiped-like shape. It has a bottom, top and four side walls. The sachet is made from a single laminated sheet with three or more layers. The innermost layer is a plastics layer that is heat-weldable and biocompatible with media being transported. “A method for manufacturing the sachet” is also disclosed.
Background for Sachets of bio-pharmaceutical liquid products
1. “1.
The present invention relates to new flexible sachets that can be used for the transport of bio-pharmaceutical products, and a manufacturing method.
2. “2.
U.S. Pat. Ser. No. No. 350080 describes rigid containers for transporting sachets that can be used as cellular culture media.
The bio-pharmaceuticals sector, taken in its broadest sense, increasingly uses flexible sachets in the 20-2,000-liter range, and in particular biocompatible sachets. These sachets are used to transport fluids in the industry such as culture media or cellular cultures, buffer solution, artificial nutrient solutions, blood products, or derived products like plasma.
Sometimes, the products in these sachets can be used thousands of kilometres away from where they were filled. These products can be extremely valuable both in terms of money and in terms of health, as they can be used in the manufacture of medication for humans. “It is important that these sachets reach their destination in a safe manner, with the original liquid inside and without contamination.
Flexible bags of this type are subjected to a variety of stresses during transport: acceleration, braking and shaking. They are subjected to many forces, including shear forces that tend to degrade the film they are made from. This is especially true at sensitive areas such as folds. These stresses often lead to the weakening, rupture, or piercing sachets.
It is important to remember that sachets containing the liquid media and products mentioned above are, by their nature, provided with multiple access ports to allow their contents to be mixed, drained, or filled. Usually, a variety of tubes will be installed in some of these ports. Tubes are often equipped with rigid materials such as clamps, valves or filters that can cause abrasion to the top of sachets during long distance transport. “A hole at the top can be as dangerous as a hole anywhere else, such as when transporting sterile materials.
This is the reason why it would be desirable to have an sachet to transport bio-pharmaceutical fluids that has a volume greater than 50 liters. This sachet should also possess all of the usual characteristics of sachets of this type, i.e. Biocompatibility, sterilizability and impermeability to gasses and oxygen in particular would be especially resistant to transport over long distances, and easy to produce.
The sachets are already known as bellows that have walls made by manually welding together three independent films.
HyClone Laboratories sells sachets like the ones above, for instance. These sachets have capacities ranging from 1 liter up to 1,000 liters. “However, due to the numerous stresses that the sachets undergo, many of them leak when they are transported long distances.
The subject of this application is therefore a flexible sachet of the bellows kind for transporting biopharmaceutical fluids of at least 50 liters volume, with four walls and a bottom, top and side wall. It is made from a single laminated sheet with three or more layers and is made preferably of four pieces.
The bio-pharmaceutical fluids that are transported include, for instance, culture media, cell cultures and buffer solutions. They can also be artificial nutrient liquids or blood products, such as plasma.
The sachet in the invention is of a bellows-type sachet. When the sachet is laid flat, it has two opposite sides folded inwards.
The term “laminated single films” is used. “The expression ‘laminated single film? “The film that is used to make the walls of a sachet appears as a single layer, but in reality it is made up of a number of layers of different films stuck together.
According to the invention, a laminated single sheet film consists of at least three layers. Preferably four layers.
The inside layer is made of a plastics material which can be heat-welded and is biocompatible to the media being transported. Polyolefins are one example of these plastics. They can also be polyethylene, especially low density and ultra-low density polyethylene.
The thickness of the layer can range from 50 m to 200m for example and more specifically between 100m and 200m.
The intermediate layer that acts as a barrier against gases like oxygen, carbon dioxide, or water vapour, can be made from polyamide (nylon), polyamide 8, polyamide 11 or polyamide 12 or copolymers of polyamide 6-6 or 6-10. You can also use mixtures of gas barrier resins, such as a polyamide or polyethylene mixture with ethylene/vinyl ethanol copolymer EVOH in polyvinylidene chloride (PVDC). Plastics that have been treated with silica or aluminum oxide can also be used. The gas barrier intermediate layer should be made from an ethylene/vinyl-alcohol copolymer under preferred implementation conditions.
The thickness of the layer can range between 6 m and 20 m for example. In particular, it is in the 10 m to 20m range.
The external layer should be made of a plastics that is heat resistant and has an insulation effect, such as polyolefin resin or polyamide resin. Polyester (PET resin) resin would be preferred.
Click here to view the patent on Google Patents.