Invented by Carlos F. Barbas, III, Hitoshi Ban, Julia Gavrilyuk, Scripps Research Institute
The Scripps Research Institute invention works as follows
A new class of diazodicarboxamides, which react efficiently and selectively to phenols as well as the phenolic side chains of tyrosine via an Ene-like process is described. This mild aqueous reaction for tyrosine modification works across a wide pH range. It expands the aqueous chemical repertoire available to modify small molecules, proteins, and peptides. The tyrosine-ligation reactions can be used to label native enzymes and antibody in buffered water. This reaction offers a new synthetic method for bispecific antibodies. This reaction is useful in the synthesis of proteins and peptides, as well as in the chemistry phenol-containing molecules.
Background for Tyrosine Bioconjugation Through Aqueous Ene-Like Reactions
Bioconjugation is the process of coupling two biomolecules together in a covalent linkage. Common types of bioconjugation chemistry are amine coupling of lysine amino acid residues (typically through amine-reactive succinimidyl esters), sulfhydryl coupling of cysteine residues (via a sulfhydryl-reactive maleimide), and photochemically initiated free radical reactions, which have broader reactivity. The product of a bioconjugation reaction is a bioconjugate. The most common bioconjugations are coupling of a small molecule (such as biotin or a fluorescent dye) to a protein, or protein-protein conjugations, such as the coupling of an antibody to an enzyme. Other less common molecules used in bioconjugation are oligosaccharides, nucleic acids, synthetic polymers such as polyethylene glycol (a.k.a. PEG a.k.a. polyethylene oxide) and carbon nanotubes.
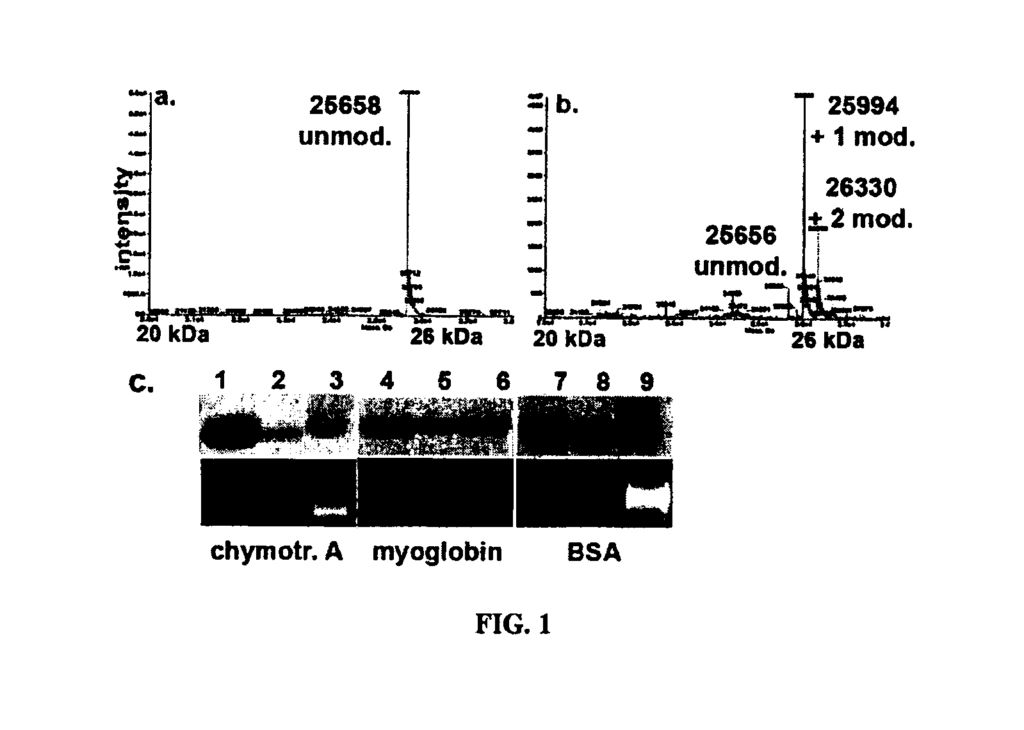
Bioconjugation relies heavily on chemoselective modification native protein functional groups. The most common functionalized amino acids are lysine and cysteine. The high concentration of lysine at the surface of proteins makes it difficult to modify specific sites. Cysteines, on the other hand, are uncommon and most commonly found in disulfide-linked pairs in proteins. This amino acid is usually labeled by reducing the disulfide target followed by a reaction with a maleimide. Recent attention has focused on the bioorthogonal modifications of the aromatic amino acids side chains of tyrosine and tryptophan. The Mannich-type additions of imines to tyrosine have been studied in mild biocompatible conditions. However, these modifications are susceptible to retro-Mannich reactions. There is a need for new bioconjugation methods that modify tyrosine in proteins.
The disclosure provides a bioconjugation orthogonal strategy that is based on the modification of tyrosine in peptides or proteins. This strategy is based on the reactivity of diazodicarboxylate-related molecules and tyrosine through an efficient aqueous Ene-like reaction. This article describes a new, efficient, and versatile tyrosine-ligation reaction. It can be used to prepare small molecules, peptides, enzymes, and antibodies.
In one embodiment, the disclosure discloses compounds with Formula I:
or a salt of the same that is pharmaceutically acceptable, wherein
When “R1 and
is a single link, and both R1 andR2 are absent when
is a dual bond;
W is an independent direct bond or O
R3 can be independently hydrogen, halogen carboxyl cyano nitro amino substituted unsubstituted, unsubstituted, alkyl substituted unsubstituted, substituted unsubstituted, aryl substituted unsubstituted, substituted unsubstituted, aryloxy substituted unsubstituted, heteroaryl substituted unsubstituted, heteroaryloxy substituted unsubstit
L can be independently H, N3, C?CHN3, C6H5, CH2CH2N3, COCH3, OCH2C??CH, OCH2COCH3, OR X?[CH2CH2Y]n? (CH2)q?N3;
X and Y each independently are CH2, O or NHCO; and
n” and “q” are both independently integers from 0-12.
In another embodiment, the disclosure provides compounds with Formula IX:
or a salt of the same that is pharmaceutically acceptable, wherein
W is an independent direct bond or O
R3 can be independently hydrogen, halogen carboxyl cyano nitro amino substituted unsubstituted, unsubstituted, alkyl substituted unsubstituted, substituted unsubstituted, aryl substituted unsubstituted, substituted unsubstituted, aryloxy substituted unsubstituted, heteroaryl substituted unsubstituted, heteroaryloxy substituted unsubstit
L can be independently H, N3, C?CHN3, C6H5, CH2CH2N3, COCH3, OCH2C??CH, OCH2COCH3, OR X?[CH2CH2Y]n? (CH2)q?N3;
X and Y each independently are CH2, O or NHCO;
n and q each independently are an integer between 0 and 12; and
R6″ is a tyrosine molecule or a residue of tyrosine in a protein or a polypeptide.