Invented by George Edwin Katibah, David Kanne, Leonard Sung, Kelsey Gauthier, Laura Hix Glickman, Justin Leong, Sarah M. McWhirter, Thomas W. Dubensky, Jr., Chinook Therapeutics Inc
The STING pathway has gained significant attention in recent years due to its involvement in various diseases, including cancer, infectious diseases, and autoimmune disorders. By targeting this pathway, researchers aim to modulate the immune response and enhance the body’s ability to fight against these conditions.
One of the main challenges in targeting the STING pathway is the development of compositions that can effectively activate STING-dependent signaling without causing excessive inflammation or immune-related adverse effects. The market for such compositions is driven by the need for safe and efficient therapies that can selectively activate the STING pathway and harness its potential for therapeutic benefit.
Pharmaceutical companies and research institutions are actively engaged in developing novel compositions that can activate the STING pathway. These compositions can be classified into various categories, including small molecules, nucleic acids, peptides, and antibodies. Each category offers unique advantages and challenges, and researchers are exploring different approaches to optimize the efficacy and safety of these compositions.
Small molecules are one of the most extensively studied categories for activating the STING pathway. These molecules can directly bind to STING and induce its activation, leading to the production of interferons and other immune-related molecules. Several small molecule candidates are currently in preclinical and clinical development, and their success could pave the way for the commercialization of STING-targeted therapies.
Nucleic acids, such as cyclic dinucleotides (CDNs), are another promising category for activating the STING pathway. CDNs can mimic the natural ligands of STING and trigger its activation. Researchers are exploring different modifications and delivery strategies to enhance the stability and bioavailability of CDNs, making them suitable for therapeutic applications.
Peptides and antibodies are also being investigated as potential compositions for activating the STING pathway. These molecules can specifically target STING or its interacting partners and modulate its activity. The development of peptide-based and antibody-based therapies offers the advantage of high specificity and reduced off-target effects.
The market for compositions targeting the STING pathway is highly competitive, with numerous companies and research institutions actively pursuing the development of novel therapies. The potential applications of STING-targeted therapies are vast, ranging from cancer immunotherapy to the treatment of viral infections and autoimmune disorders. As a result, the market is expected to witness significant growth in the coming years.
In conclusion, the market for compositions for activating the STING-dependent signaling is rapidly expanding as researchers and pharmaceutical companies recognize the therapeutic potential of targeting this pathway. The development of safe and effective compositions that can selectively activate the STING pathway is a key focus for researchers, and various categories of compositions, including small molecules, nucleic acids, peptides, and antibodies, are being explored. With the potential to revolutionize the treatment of various diseases, the market for STING-targeted therapies is poised for significant growth in the near future.
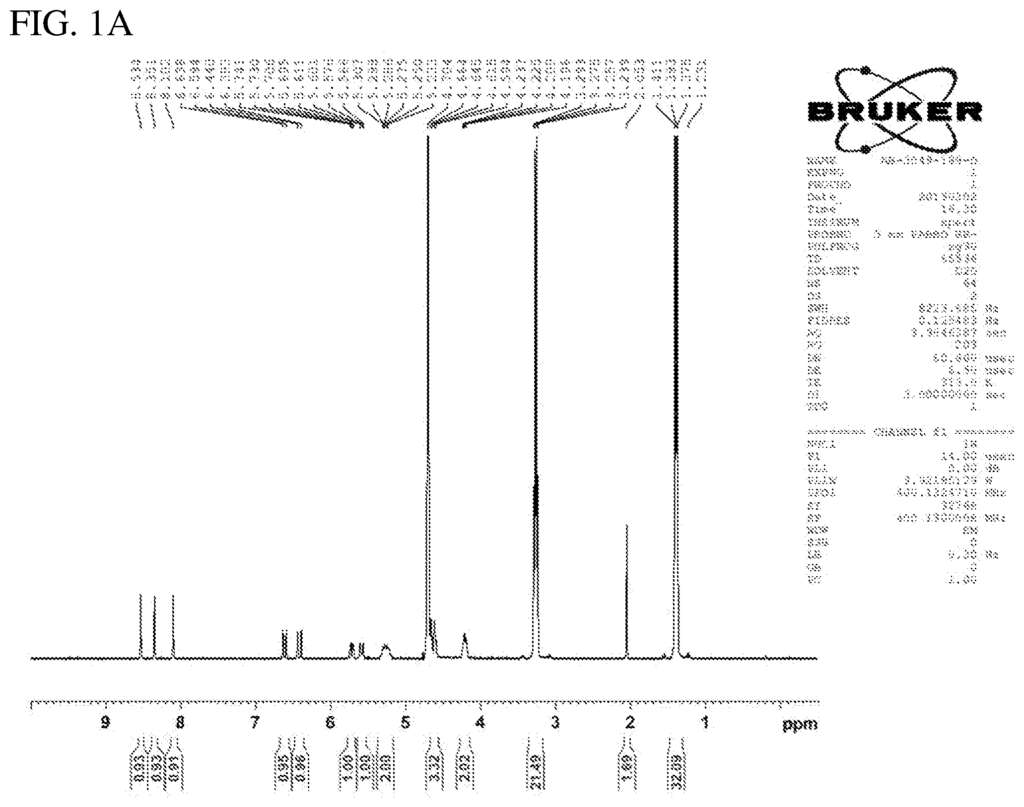
The Chinook Therapeutics Inc invention works as follows
The present invention provides highly effective cyclic di-nucleotide immune stimulators which activate DCs through a newly discovered cytoplasmic STING receptor. The CDNs are provided as a composition containing one or more cyclic dinucleotides that induce type I human interferon production dependent on STING. CDNs and, most preferably, one or more bis-3?5,5?CDNs that are 2?-fluoro substituted, bis-3?5,5?
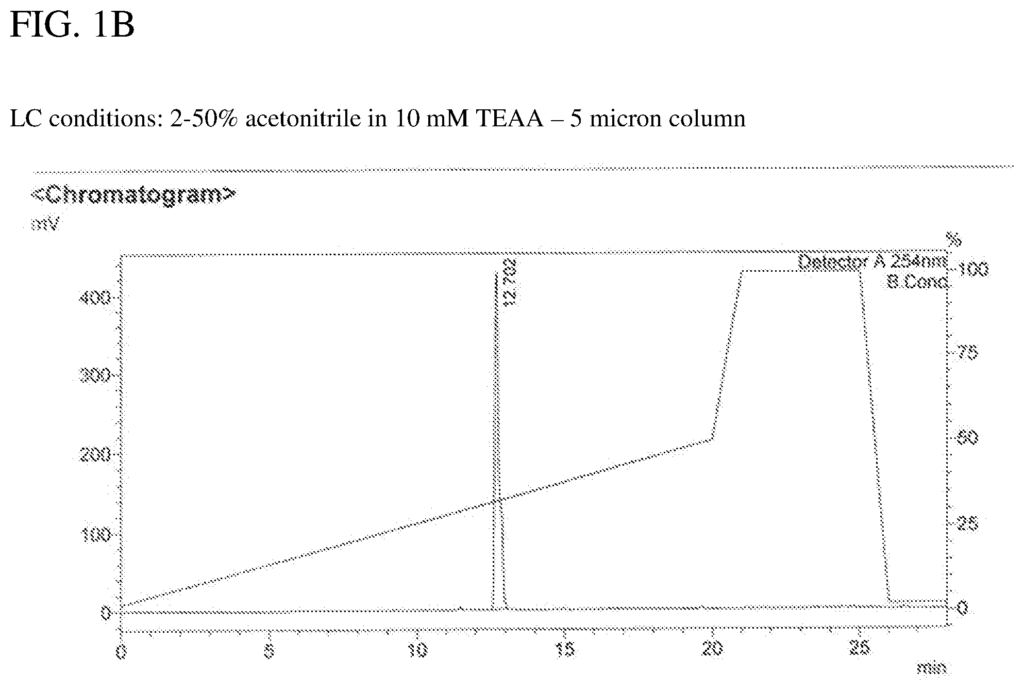
Background for Compositions for activating the?stimulator interferon gene 13 dependent signalling
The following discussion is provided to help the reader understand the invention. It is not intended to describe prior art or to constitute it.
New insights into immune-evasion mechanisms, along with combination treatment regimens which potentiate or boost the potency therapeutic vaccination, either directly or indirectly, through combination with immune-checkpoint inhibitors or another therapy, have served as the basis for the creation of vaccines or immunomodulators that can prime, or boost, an effective adaptive immunity response consisting of cancer-specific CD4+ or CD8+ cells specific for targeted malignancies, resulting in a clinical benefit and antitumor effect. The way in which the innate immune response is shaped by the targeted ligands can be used to design vaccines and immunomodulators. (Reed et al. Trends Immunol. 30: 23-32 2009; Dubensky & Reed, Semin. Immunol., 22: 155-61, 2010; Kastenmuller et al., J. Clin. Invest., 121: 1782-1796, 2011; Coffman et al., Immunity, 33: 492-503, 2010).
STING is an adaptor protein in the cytoplasm of host mammalian cells that activates the TANK binding kinase (TBK1)?IRF3 and the NF-?B signaling axis, resulting in the induction of IFN-?. STING is a protein adaptor in the cytoplasm mammalian host cells that activates the TANK-binding kinase TBK1?IRF3?NF-?B-signaling axis. This results in the induction IFN-? STING and other gene products strongly activate innate immune system. STING, as a component of host cytosolic security (Vance, et. al., 2009), senses intracellular pathogens, and then induces production of IFN?. This leads to an adaptive pathogen-specific immunity response, consisting of antigen-specific T cells, CD4 and/or CD8 and pathogen specific antibodies. U.S. Patent. Nos. Nos. Med. Med. 18: 5631 (2008).
The enzyme responsible for the production of STING-binding CDNs within mammalian cell was identified as an uncharacterized mouse gene that shares significant structural similarities with the human oligoadenylate synase cyclic GMP AMP synthase. Sun et al., Science 339(6121):786-91, 2013. This enzyme, called cyclic GMP AMP Synthase(cGAS), catalyzes cGAMP synthesis from ATP and GTP when DNA is present. This cGAMP is then used as a second message that activates and binds STING. The cGAS produced CDNs are structurally different from the bacterially-produced CDNs because they have an unusual phosphodiester bond. While the CDNs produced by bacteria contain a bis-3,5? The mammalian CDNs, however, contained a 2?,5-? Mammalian CDNs contained one 2?,5-? Linkage or so-called “mixed linkage” (ML), or non-canonical CDNs. These 2?-5?-3?-5? molecules bind STING with nM affinity, about 300-fold better than bacterial c-diGMP.” These 2?,5?-3?,5 molecule bind STING at nM affinity. This is 300-fold more than bacterial di-GMP.
Human STING(hSTING) has also known polymorphisms including alleles that encode histidine in position 232. These are resistant to bis-3? “Human STING (hSTING) also has known polymorphisms, including alleles encoding histidine at position 232, which are refractory to bis-3?,5-? Non-canonical CDNs with mixed linkage (Diner and al. Cell Reports 3: 1355-61 (2013); Jin and al. Genes and Immunity 12: 263-69 (2011)). The hSTING gene has been shown to have single nucleotide variations that affect the response to canonical CDNs derived from bacteria (Diner and al. 2013; Gao et. al. Cell 154: 748-762; Conlon and al. al., J. Immunol. 190: 5216-5225, 2013). Five haplotypes (WT; REF; HAQ; AQ and Q) of hSTING were reported, which differ at amino acid positions (71, 230 232, and 293) (Jin et. al. 2011; Yi Yi et. al. PLOS One 8, e77846, 2013,). Cells that express hSTING are reported to respond poorly to stimulation by bacterial CDNs, cGAMP, C-diAMP and cDiGMP with bis-(3 or 5?) linkages. Cells expressing hSTING reportedly respond poorly to stimulation with bacterial CDNs cGAMP, c-di-AMP and c-di GMP having bis(3?, 5?) It has been suggested that 2?,5-3?,5 molecules are more potent ligands for hSTING (Zhang et al., Mol. Cell. Cell. Cell. Cell 51:135-39 (2013).
It is the object of this invention to provide compositions or methods that modulate immune response to diseases. The invention also aims to provide compositions or methods that provide cyclic dinucleotide analogues with improved properties when used for activation mammalian STING, preferably human. “It is another object of the present invention to provide compositions for cancer treatment.
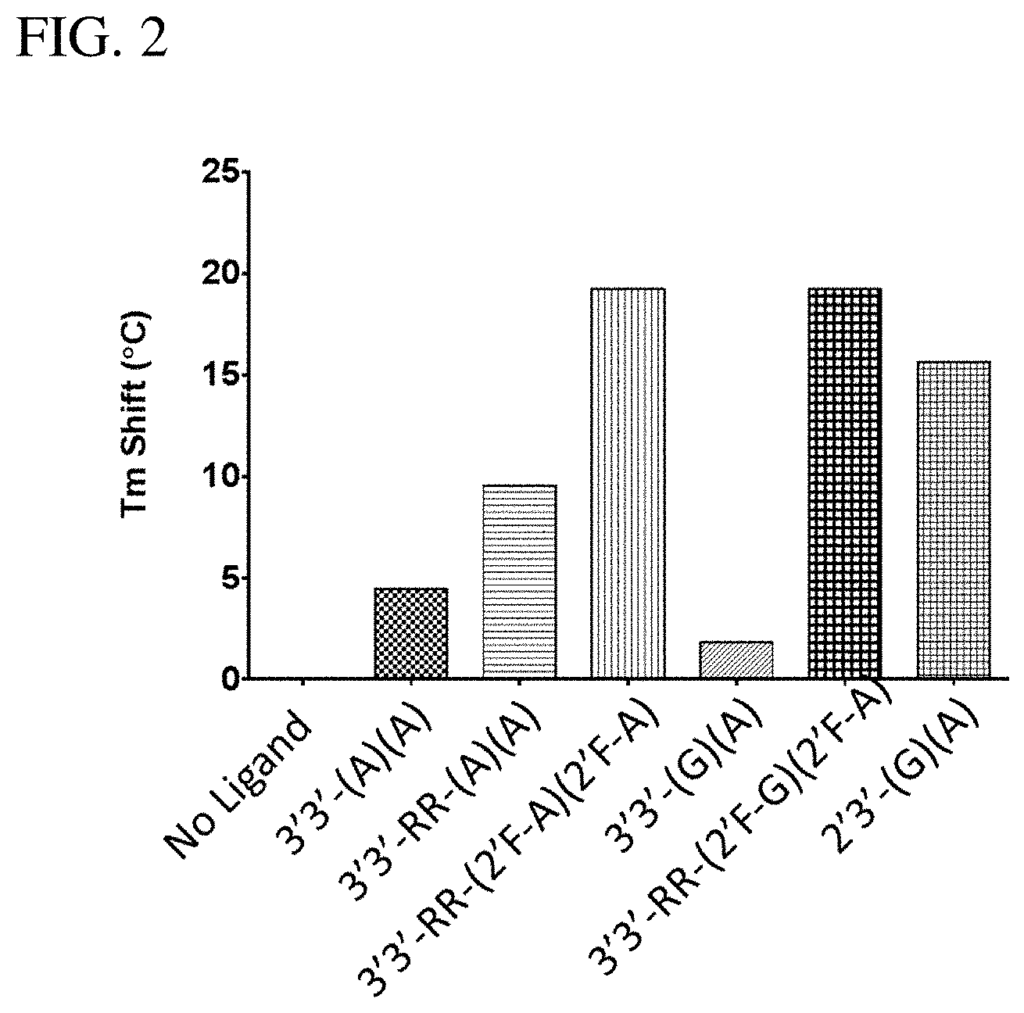
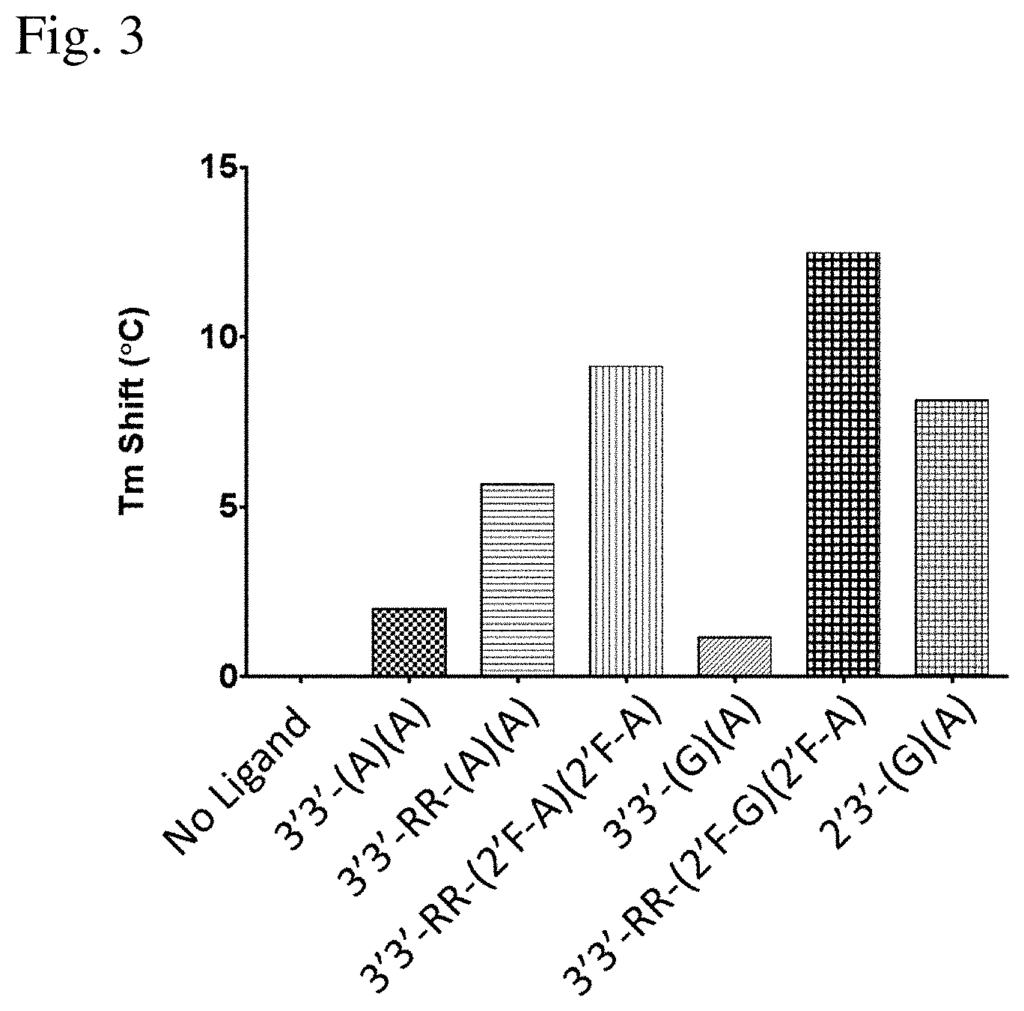
In a first aspect of the invention, compositions are provided that contain: one or two mono- or bi-fluoro substituted Bis-3?,5? “In a first aspect, the present invention provides compositions comprising: one or more mono- or difluoro substituted bis-3?,5? The cyclic purine dinucleotides (?mono- or di-F CDN compounds? STING? c-di-GMP (i.e. 3?3?-(G)(G)), bis-3?,5? c-di-AMP (i.e. 3?3-(A/A) or CDA, or bis-3?5,5? c-GMP-AMP (i.e. When measured with at least one STING human allele (h), 3?3-(G)?(A) or CGAMP is used. This is preferably measured using the hSTING (REF) allele as described by Ishikawa H. and Barber G. N. 2008 Nature 455, 675-678. The protein sequence is NCBI Reference Sequence NC_938023. (SEQ ID No: 1; FIG. 6). This is best measured in vitro, using a mammalian line (e.g. HEK293T) that does not express an endogenous functional STING, but which has been modified so as to express the hSTING (REF) allele stably, as described below (e.g. See Example 12.
In a related aspect of the invention, compositions are provided that include: one or two mono- or diF-CDNs that bind to at least one allelic human protein product STING with an affinity greater than 10 times higher than any one or both bis-3? c-di-GMP (i.e. 3?3?-(G)(G)), bis-3?,5? c-di-AMP (i.e. 3?3-(A),(A), or CDA, or bis-3?5,5? c-GMP-AMP (i.e. When measured with at least one human protein, 3?3??-(G?)(A)or cGAMP. This is best measured by using the isolated protein encoded the hSTING (REF) allele as described in Ishikawa, H. and Barber G. N. Nature 455, 675-678), whose protein sequence is NCBI Reference Sequence NC_938023, using a differential scanning fluorometry method as described below (e.g. “See Example 11.
As described below, substituting one or both of 2?-hydroxyls on the bis-3,5? The cyclic Purine Dinucleotides in the present invention have a significant improvement in their ability to bind human STING. In the present invention, a number of CDNs that are 2?-fluoro substituted are used. The preferred 2?fluoro-substituted CDNs are those derived from c?di?AMP, c?di?GMP or c?di?IMP by substituting one or both of the 2?-hydroxyls. This list should not be considered exhaustive. “Hereafter, additional aspects and embodiments for mono- or bi-F-CDN are described.
In a second aspect of the invention, it provides a 3?,5?-3??,5? cyclic purine dinucleotide (mono- or di-F-CDN) compound of Formula I: “In a second aspect, the present invention provides a mono- or difluoro substituted 3?,5?-3??
Or a prodrug or a pharmaceutically accepted salt, pharmaceutically acceptable solvate, or pharmaceutically acceptable hydrate thereof wherein each of R1 andR2 is independently purine; each of R3 & R4 is independently F, H, OH, or F provided that either one or both R3 & R4 are F. Each R5 & R6 is independent OH orSH.
In certain embodiments, R5 and R6 are each SH. When each R5 or R6 is SH in preferred embodiments, the compositions contain one or more stereoisomers that are substantially pure, such as Sp,Sp or Rp/Rp. In certain embodiments, the compositions contain one or more Rp,Rp monoisomers.
The following general structure of purines R1 and R2 may be used in the compounds described below:
Each Each R Each Each
In certain embodiments, R1 and R2 can be independently selected from a group of adenine or guanine. In certain embodiments, R1 and/or R2 can be independently adenine, guanine or both. In some embodiments one R1 or the other R1 is adenine and the other R1 is guanine. Each of R1 is adenine or Guanine. In a preferred form, R1 or R2 can be independently adenine and guanine.
In some embodiments, R5 and each R6 are SH and R1 is independently:
R15 and R16 can be independently H or C(O)R, while R? R? “In some embodiments R15 can be H or C(O),-isopropyl, and R16 can be H or C(O),-phenyl.
In certain embodiments of the second and embodiments thereof, the compound of Formula I may be selected from a group consisting:
Or a prodrug or a pharmaceutically accepted salt, pharmaceutically accept solvate, or pharmaceutically accept hydrate thereof.
In preferred embodiments, R3 and R4 are each F. This is herein referred to as 2?,2′-diF, or di-2?F.
In some embodiments the one or more compounds are 2?,2?-diF-Rp,Rp cyclic purine dinucleotides (thus in which each R5 and R6 is SH), including, but not limited to, 2?,2?-diF-Rp,Rp-c-di-AMP (also referred to herein as 2?,2?-diF-R,R-CDA or 3?3?-RR-(2?F-A)(2?F-A)), 2?,2?-diF-Rp,Rp-c-di-GMP (also referred to herein as 2?,2?-diF-R,R-CDG or 3?3?-RR-(2?F-G)(2?F-G)), 2?,2?-diF-Rp,Rp-c-di-IMP (also referred to herein as 2?,2?-diF-R,R-CDI or 3?3?-RR-(2?F-I)(2?F-I)), 2?,2?-diF-Rp,Rp-c-AMP-GMP (also referred to herein as 2?,2?-diF-R,R-cGAMP or 3?3?-RR-(2?F-G)(2?F-A)), 2?,2?-diF-Rp,Rp-c-AMP-IMP (also referred to herein as 2?,2?-diF-R,R-cIAMP or 3?3?-RR-(2?F-I)(2?F-A)), 2?,2?-diF-Rp,Rp-c-GMP-IMP (also referred to herein as 2?,2?-diF-R,R-cGIMP or 3?3?-RR-(2?F-G)(2?F-I)), and analogs thereof.

Click here to view the patent on Google Patents.
Leave a Reply