Invented by Jackie SHENG, Bo Liu, Gensun Biopharma Inc
Trispecific antagonists are designed to bind to three different targets, typically receptors or proteins involved in disease processes. By simultaneously blocking multiple pathways, these drugs can effectively disrupt disease progression and provide a more comprehensive treatment approach compared to traditional monospecific antagonists.
One of the key advantages of trispecific antagonists is their ability to target complex diseases with multiple underlying mechanisms. For example, in cancer treatment, trispecific antagonists can simultaneously inhibit tumor growth, angiogenesis, and immune evasion pathways, leading to a more robust and effective anti-cancer response. This multi-targeted approach has the potential to overcome drug resistance and improve patient outcomes.
The market for trispecific antagonists is particularly promising in the field of immunotherapy. By targeting multiple immune checkpoints, these drugs can enhance the anti-tumor immune response and improve the efficacy of existing immunotherapies such as immune checkpoint inhibitors. Trispecific antagonists can also be engineered to selectively activate immune cells, further enhancing their therapeutic potential in cancer treatment.
In addition to cancer, trispecific antagonists hold promise in other disease areas such as autoimmune disorders, inflammatory diseases, and infectious diseases. By targeting multiple disease pathways, these drugs can modulate the immune response and provide a more comprehensive treatment approach. This potential has attracted the attention of pharmaceutical companies and biotech startups, leading to a surge in research and development activities in this field.
The market for trispecific antagonists is also driven by the increasing availability of advanced technologies for drug discovery and development. Advances in protein engineering, computational biology, and high-throughput screening have facilitated the design and optimization of trispecific antagonists with improved potency, selectivity, and stability. These technological advancements have accelerated the development timelines and reduced the costs associated with bringing trispecific antagonists to market.
However, there are still challenges to be addressed in the development and commercialization of trispecific antagonists. The complexity of designing and manufacturing these drugs, as well as the potential for off-target effects, require careful optimization and thorough preclinical and clinical evaluation. Regulatory considerations and reimbursement policies also play a crucial role in determining the market access and commercial success of trispecific antagonists.
Despite these challenges, the market for trispecific antagonists is expected to grow significantly in the coming years. The potential of these drugs to provide a more comprehensive treatment approach and improve patient outcomes makes them an attractive option for pharmaceutical companies and healthcare providers. As research and development efforts continue to advance, trispecific antagonists have the potential to revolutionize the treatment landscape across various disease areas, offering new hope for patients and transforming the future of medicine.
The Gensun Biopharma Inc invention works as follows
Antitumor antagonists are disclosed that bind to components of angiogenesis pathways, components of TGF pathway and/or immune checkpoint regulator. A method for treating proliferative diseases with antitumor inhibitors is also disclosed.
Background for Trispecific antagonists
The host’s inability to eliminate cancerous cells remains a significant problem.” The use of monoclonal therapeutic antibodies to treat cancer is on the rise. However, resistance to these antibodies has been observed due to the different molecular pathways that underlie cancer growth and metastasis. Cancer cells are able to counteract the immune surveillance, despite the fact that the immune system is the primary mechanism for cancer prevention. There is a need to develop improved therapeutic antibodies or antagonists, and ways of treating chronic viral infections and cancer with these reagents.
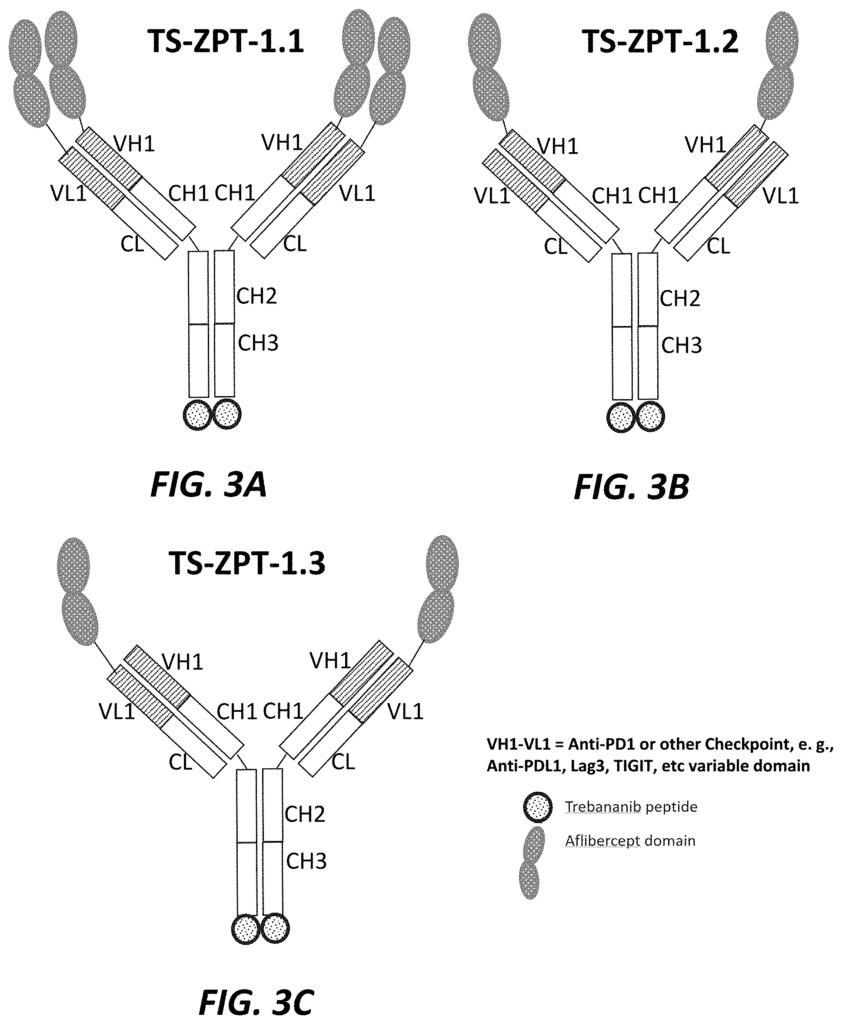
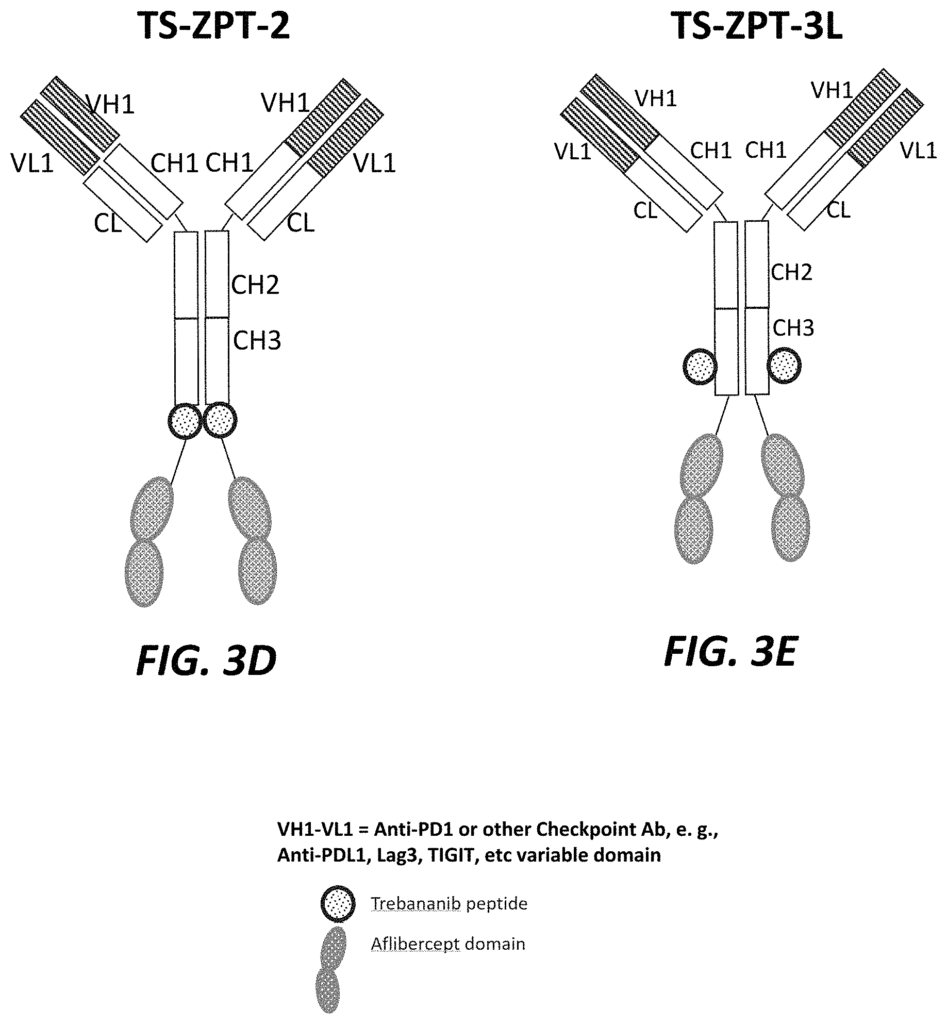
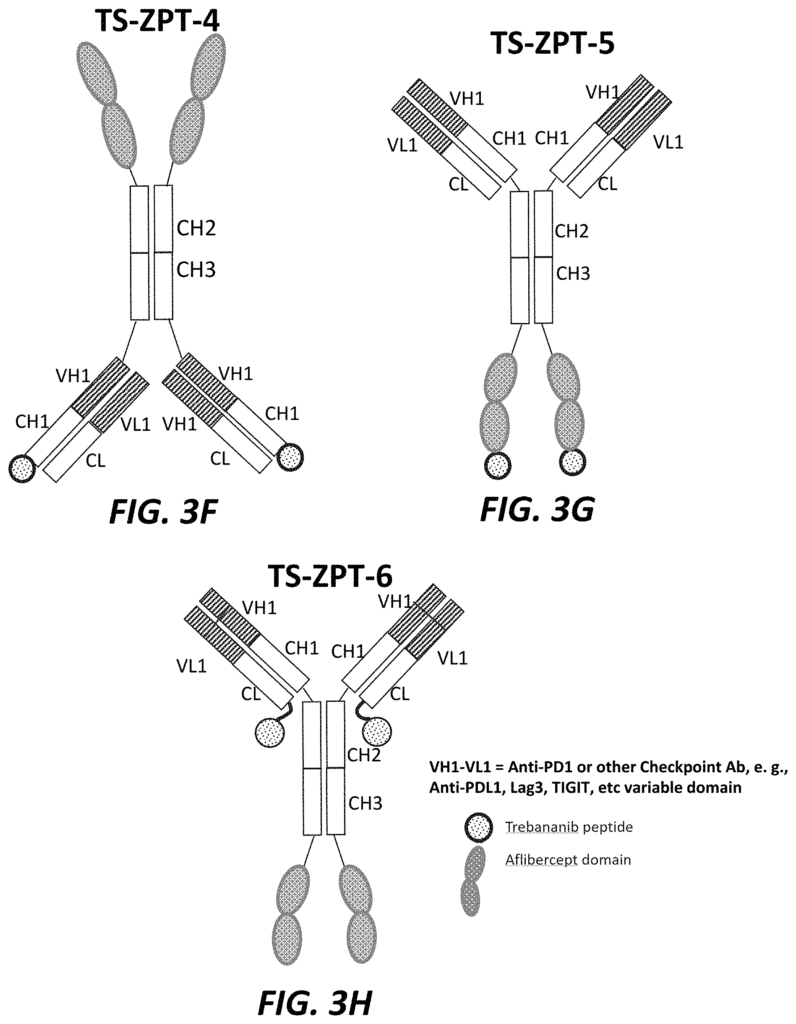
The present application relates in part to a trispecific inhibitor, which includes: an immunoglobulin framework comprising a PD-1 domain, a PD-L1 domain, a TIGIT domain, and an anti-LAG-3 domain; a targeting second domain that binds to VEGF, and contains one or more peptides derived form VEGFR, and a targeting third domain that comprises a peptide inhibiting the Tie-2 signaling pathway.
The present application also relates to a trispecific inhibitor, which comprises: an immunoglobulin-scaffold containing a PD-1 variable, a PD-L1-variable, a TIGIT-variable, and an antiLAG-3-variable domain; a second-targeting domain consisting of anti PD-1-variable domains as well as anti PD-L1-variable domains. A third-targeting domain consists of a peptide peptide peptide a
Another aspect of the current application relates to a Trispecific Antagonist, which comprises: An immunoglobulin-based scaffold consisting of a CH1-domain, a? The present application also relates to a trispecific antagonist, comprising: an immunoglobulin scaffold consisting of a CH1 domain, a CH2 domain and a CH3domain; a first targeting domain consisting of one or more variable domains selected from the group consisting of anti-VEGF variable domains; a second targeting domain comprising a TGF-?
The present application also relates to a trispecific inhibitor that includes an immunoglobulin-based scaffold having a CH1-domain, a PD-L1-domain, a anti-TIGIT-domain, and a LAG-3-domain, a first-targeting domain comprising a variable domain selected from a group consisting anti-PD-1-variable domains, anti PD-L1-variable domains, anti TIGIT-variable domains and anti LAG-3-domain, a a a a a a a a a a a a a a a – a a a – a a – a a-, a-, a-, The second targeting is structurally connected to the carboxy-terminal CH3 domain. And the third targeting is structurally attached to the carboxy-terminal second targeting.
Another aspect of the current application relates a bispecific inhibitor that comprises an immuneglobulin scaffold consisting of a PD-1 variable, a PD-L1 or anti-TIGIT domain, and a VEGFR component, and a third targeting domain structurally linked to the carboxy-terminal portion of the second targeting.
The present application also includes a method of treating a disorder of cell proliferation. The method consists of giving a trispecific antagonist of the present application to a patient in need.
Definitions
The terms “technical and scientific” used in this document have the same meaning as they would be understood by a person of ordinary skill in the field to which the disclosed compositions and methods belong. As used in this document and the accompanying claims, singular forms such as?a?,? ?an,? The terms?an?,? Plural references are acceptable unless it is clear that the context requires otherwise. For example, a reference to “a peptide” is acceptable. This includes “one or more” peptides. Includes ‘one or more? These peptides or a?plurality? “With respect to the teachings of the present application any published patent or application described in this application are expressly incorporated herein by reference.
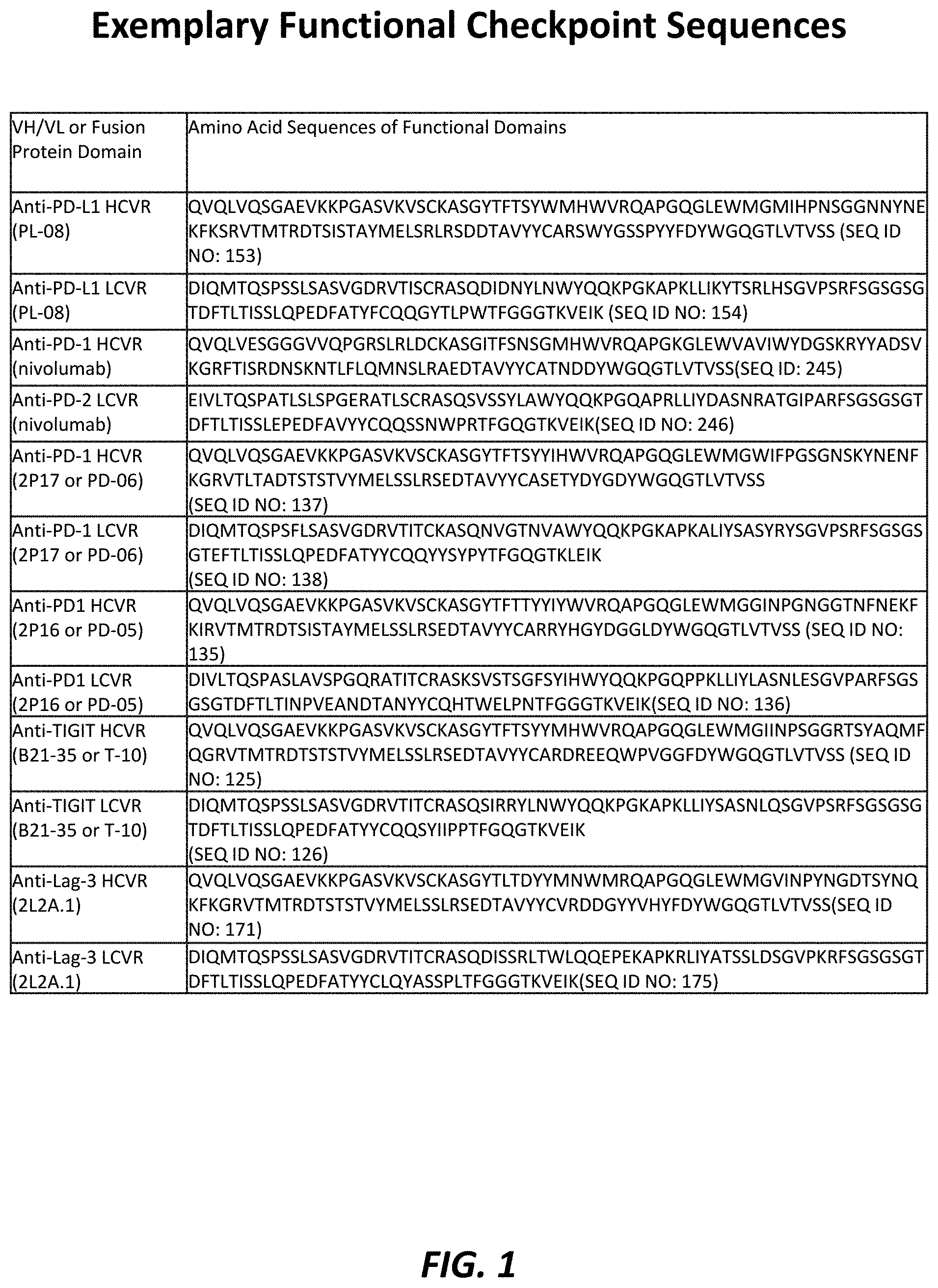
As used in this document, the term ‘PD-1’ refers to any form of PD-1 and variants thereof that retain at least part of PD-1?s activity. Refers to all forms of PD-1, and variants of PD-1 that retain at least a part of its activity. Unless otherwise indicated, such as through a specific reference to human PD-1 or by referring to all mammalian native sequence PD-1 species, e.g. human, canine, feline, equine, and bovine. Below is an example of a human PD-1 sequence.
(SEQ?ID?NO:?180)\nMQIPQAPWPVVWAVLQLGWRPGWFLDSPDRPWNPPTFSPALLVVTEGDNA\nTFTCSFSNTSESFVLNWYRMSPSNQTDKLAAFPEDRSQPGQDCRFRVTQL\nPNGRDFHMSVVRARRNDSGTYLCGAISLAPKAQIKESLRAELRVTERRAE\nVPTARPSPSPRPAGQFQTLVVGVVGGLLGSLVLLVWVLAVICSRAARGTI\nGARRTGQPLKEDPSAVPVFSVDYGELDFQWREKTPEPPVPCVPEQTEYAT\nIVFPSGMGTSSPARRGSADGPRSAQPLRPEDGHCSWPL.
As used in this document, the term ‘PD-L1’ refers to any form of PD-L1 and variants thereof that retain at least part of PD-L1 activity. Refers to any form or variant of PD-1 that retains at least a part of PD-1’s activity. Unless otherwise indicated, such as by a specific reference to human PDL1, PDL1 refers to all mammalian native sequence PDL1 species, e.g. human, canine (including feline), equine (including bovine), and feline. Below is an example of a human PD-L1 sequence.
(SEQ?ID?NO:?181)\nMRIFAVFIFMTYWHLLNAFTVTVPKDLYVVEYGSNMTIECKFPVEKQLDL\nAALIVYWEMEDKNIIQFVHGEEDLKVQHSSYRQRARLLKDQLSLGNAALQ\nITDVKLQDAGVYRCMISYGGADYKRITVKVNAPYNKINQRILVVDPVTSE\nHELTCQAEGYPKAEVIWTSSDHQVLSGKTTTTNSKREEKLFNVTSTLRIN\nTTTNEIFYCTFRRLDPEENHTAELVIPELPLAHPPNERTHLVILGAILLC\nLGVALTFIFRLRKGRMMDVKKCGIQDTNSKKQSDTHLEET.
As used in this document, the term “TIGIT” refers to any form of TIGIT and variants thereof that retain at least part of its activity. Refers to all forms of TIGIT, and variants of TIGIT that retain at minimum part of TIGIT’s activity. TIGIT is a term that includes all mammalian native sequences of TIGIT unless otherwise indicated, for example, by referring to human TIGIT. This would include human, canine and feline TIGIT as well as equine and bovine TIGIT. This is a typical human TIGIT sequence.
(SEQ?ID?NO:?179)\nMTGTIETTGNISAEKGGSIILQCHLSSTTAQVTQVNWEQQDQLLAICNAD\nLGWHISPSFKDRVAPGPGLGLTLQSLTVNDTGEYFCIYHTYPDGTYTGRI\nFLEVLESSVAEHGARFQIPLLGAMAATLVVICTAVIVVVALTRKKKALRI\nHSVEGDLRRKSAGQEEWSPSAPSPPGSCVQAEAAPAGLCGEQRGEDCAEL\nHDYENVLSYRSLGNCSFFTETG.
The term “LAG-3” is used in this document. refers to any form of LAG-3 and variants thereof that retain at least part of the activity of lymphocyte-activation gene 3 (LAG-3). LAG-3 is the native sequence of all mammalian species, including human, canine and feline. This is a typical human LAG-3 sequence.
(SEQ?ID?NO:?293)\nMWEAQFLGLLFLQPLWVAPVKPLQPGAEVPVVWAQEGAPAQLPCSPTIPL\nQDLSLLRRAGVTWQHQPDSGPPAAAPGHPLAPGPHPAAPSSWGPRPRRYT\nVLSVGPGGLRSGRLPLQPRVQLDERGRQRGDFSLWLRPARRADAGEYRAA\nVHLRDRALSCRLRLRLGQASMTASPPGSLRASDWVILNCSFSRPDRPASV\nHWFRNRGQGRVPVRESPHHHLAESFLFLPQVSPMDSGPWGCILTYRDGFN\nVSIMYNLTVLGLEPPTPLTVYAGAGSRVGLPCRLPAGVGTRSFLTAKWTP\nPGGGPDLLVTGDNGDFTLRLEDVSQAQAGTYTCHIHLQEQQLNATVTLAI\nITVTPKSFGSPGSLGKLLCEVTPVSGQERFVWSSLDTPSQRSFSGPWLEA\nQEAQLLSQPWQCQLYQGERLLGAAVYFTELSSPGAQRSGRAPGALPAGHL\nPAGHLLLFLILGVLSLLLLVTGAFGFHLWRRQWRPRRFSALEQGIHPPQA\nQSKIEELEQEPEPEPEPEPEPEPEPEPEQL.
The term “agonist” refers to a substance that promotes (i.e., induces, causes or increases) the biological activity or effect of another molecule. A substance that promotes the biological activity of another molecule (i.e. induces, causes or enhances it) is called an agonist. The term agonist includes substances that bind receptors, such as antibodies, and substances that promote receptor function (e.g. by activating a protein associated with the receptor) without binding to them.
The term “anti-” or “inhibitor” is used. “The term?inhibitor’ or?antagonist? Refers to a substance which prevents, blocks or inhibits the biological activity or effects of another molecule such as a ligand or receptor. “An antagonist can be either a monospecific antibody, bispecific antibody or trispecific antibody.
As used in this document, the term “antibody” is defined as: Refers to a complex polypeptide that binds to and recognizes an antigen via one or more immunoglobulin-variable regions. A whole antibody can be an antibody binding fragment, or just a single chain. The term “antibody” is used. The term?antibody’ encompasses a variety of broad classes that can be differentiated biochemically. Heavy chains can be classified by those who are skilled in the field as alpha-delta-epsilon-gamma and mu or?. The artful will recognize that heavy chains are classified as alpha, delta, epsilon gamma and mu or? There are some subclasses (e.g.?1-?4). The nature of the chain determines the class. The nature of this chain determines the?class? The subclasses of immunoglobulins (isotypes), e.g. IgG1, IgG2, IgG3, IgG4, and so on. They are well-characterized and known to confer functional specialty. The skilled artisan can readily distinguish modified versions of each of these isotypes and classes in light of the present disclosure, and are, therefore, within the scope and application of the current disclosure. The present disclosure covers all immunoglobulin classes. However, this discussion will be primarily focused on the IgG class.
Antibodies and antibody antagonists” of the present application include, but aren’t limited to: polyclonal antibodies, monoclonal antibodies, monospecific antibodies, bispecific antibodies, trispecific antibodies, humanized or primatized antibodies, chimeric, single chain, and chimeric-chimeric chains. The antibodies disclosed in this document may come from any animal, including birds and mammal. The antibodies should be human, murine antibodies, donkey antibodies, rabbit antibodies, goat antibodies, guinea-pig antibodies, horse antibodies, llama antibodies, and chicken antibodies. The variable region in some embodiments may be derived from condricthoids (e.g. sharks).
Click here to view the patent on Google Patents.
Leave a Reply