Invented by Claire M. Fraser, Mark D. Adams, Jeannine D. Gocayne, Clyde A. Hutchison, III, Hamilton O. Smith, J. Craig Venter, Owen White, University of North Carolina at Chapel Hill, Johns Hopkins University, J Craig Venter Institute Inc
The nucleotide sequence of the Mycoplasma genitalium genome has been fully sequenced, and this has led to the identification of potential targets for new drugs and vaccines. The market for this sequence is driven by the need for new diagnostic tools and treatments for Mycoplasma genitalium infections.
The market for fragments of the Mycoplasma genitalium genome is also growing. These fragments can be used to develop diagnostic tests that can detect the presence of the bacterium in clinical samples. This is important because Mycoplasma genitalium infections are often asymptomatic, and therefore, it can be difficult to diagnose the infection.
The uses of the Mycoplasma genitalium genome sequence and its fragments are diverse. They can be used to develop new drugs and vaccines, as well as diagnostic tests. They can also be used to study the biology of the bacterium and its interactions with the host.
The market for nucleotide sequence of the Mycoplasma genitalium genome, fragments thereof, and uses thereof is driven by the increasing prevalence of Mycoplasma genitalium infections and the need for new diagnostic tools and treatments. The market is expected to grow in the coming years as more research is conducted on this bacterium and its interactions with the host.
In conclusion, the market for nucleotide sequence of the Mycoplasma genitalium genome, fragments thereof, and uses thereof is growing rapidly. This market is driven by the need for new diagnostic tools and treatments for Mycoplasma genitalium infections. The uses of the Mycoplasma genitalium genome sequence and its fragments are diverse, and they have the potential to lead to the development of new drugs and vaccines. As more research is conducted on this bacterium, the market for its genome sequence and fragments is expected to grow further.
The University of North Carolina at Chapel Hill, Johns Hopkins University, J Craig Venter Institute Inc invention works as follows
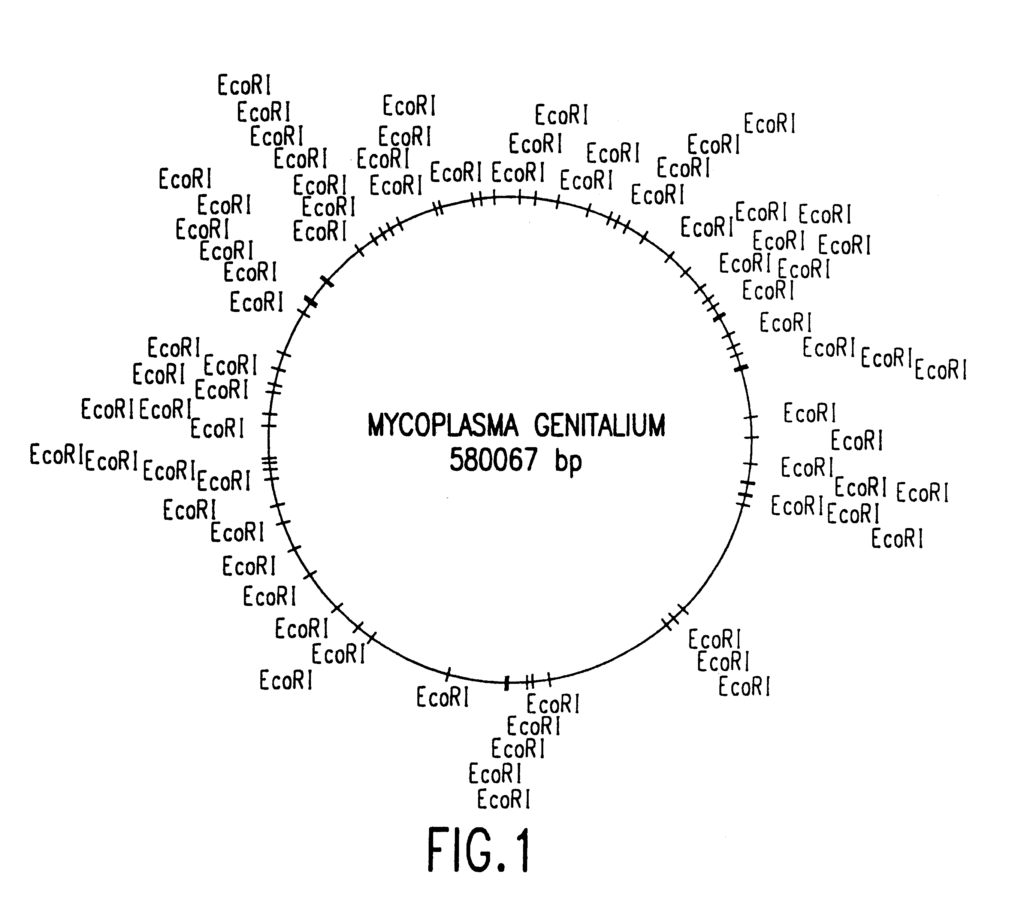
The present invention provides the nucleotide sequencing of the entire genome for Mycoplasma Genitalium. SEQ ID No:1. The present invention also provides sequence information on computer-readable media and computer-based methods and systems that facilitate its use. The present invention, in addition to the complete genomic sequence, identifies the protein-encoding segments of the genome and identifies any regulatory elements that modulate the expression.
Background for Nucleotide Sequence of the Mycoplasma Genitalium Genome, Fragments thereof, and Uses Thereof
1. “1.
The present invention is in the field of Molecular Biology. The invention discloses compositions containing the nucleotide sequencing of Mycoplasma Genitalium or fragments thereof and their use in medical diagnosis, therapies, and pharmaceutical development.
2. Related Background Art
Mycoplasmas is the smallest known free-living bacteria (Colman, S. D. and others, Mol. Microbiol. 4:683-687 (1990)). It is believed that mycoplasmas evolved from higher-gram-positive bacteria by losing genetic material (Bailey C. C. and al., J. Bacteriol. 176:5814-5819 (1994)). Mycoplasma genitalium is widely considered the smallest self replicating biological system. The molecular size (570-600 kp) of its genome (Pyle L. E. et.al., Nucleic Acids Res. 16(13):6015-6025 (1988); Peterson, S. N. et al., J. Bacteriol. 175:7918-7930 (1993)). All mycoplasmas are characterized by a lack of a cell wall, small genomes, and low G+C contents (Razin S., Microbiol. Rev. 49(4):419-455 (1985); Peterson, S. N. et al., J. Bacteriol. 175:7918-7930 (1993)). Some mycoplasmas such as M genitalium have a specialized code usage whereby UGA encodes the amino acid tryptophan instead of serving as a stopping codon. (Inamine, J. M. et. al., J. Bacteriol. 172:504-506 (1990); Tanaka, J. G. et al., Nucleic Acids Res. 19:6787-6792 (1991); Yamao, F. A. et al., Proc. Natl. Acad. Sci. USA 82:2306-2309 (85 )).”)
Mycoplasmas have long been recognized as significant pathogens in humans, animals and plants (Bailey C. C., et. al., J. Bacteriol. 176:5814-5819 (1994)). Mycoplasmas have metabolic systems that are biosynthetically lacking. They depend on their host’s microenvironment to get essential precursor molecules such as amino acids, fat acids and sterols. (Baseman, J. B., 1987. Micoplasma Membranes Vol. 20. The Plenum Press New York, N.Y .).
In particular M. genitalium is a newly-discovered species. It was first isolated from 1980 in the urethras and urethras o human males with nongonococcal Urethritis. (Tully J. G. J. Syst. Bacteriol. 33:387-396 (1983)). In specimens from pneumonia patients, M. genitalium was also identified as a coisolate of Micoplasma pneumoniae (Baseman J. B. et al., J. Clin. Microbiol. 26:2266-2269 (1988)). Infections with M. genitalium opportunistic have been reported in HIV-1-infected individuals (Lo, S.C. et al. J. Trop. Med. Hyg. 41:601-616 (1989); Lo, S.-C. et al., Amer. J. Trop. Med. Hyg. 41:601-616 (1989); Sasaki, Y. et al., AIDS Res. Hum. Retrov. 9(8):775-780 (1993)). Mycoplasmas may also induce cytokines such as tumor necrosis factors, which can enhance HIV replication. Biophys. Res. Commun. 170:1365-1370 (1990)).
A high homology of amino acids exists between the attaching protein of M. genitalium, and the aligned HLA proteins of some human Class II major Histocompatibility Complex proteins, suggesting that M. genitalium may play an important part in triggering autoimmune processes, thereby aggravating immunodeficiency symptoms of acquired immune deficiency (AIDS).” (Montagnier L. et.al., C.R. Acad. Sci. Paris 311(3):425-430 (1990); Root-Bernstein, R. S. et al., Res. Immunol. 142:519-523 (1991); Bisset, L. R. Autoimmunity 14:167-168 (1992)). The development of a diagnostic immunoassay to detect M. genitalium infections using monoclonal antigens specific for certain M.genitaliumantigens. Baseman, J. B. et al., U.S. Pat. No. 5,158,870.
Due to its small genome size, M. Genitalium is a good model for determining how many genes and proteins are required for an independent existence. M. genitalium has a low G+C and base pair content. This, along with the UGA tryptophan code, has made it difficult to sequence using conventional techniques. Rev. 49(4):419-455 (1985); Colman, S. D. et al., Gene 87:91-96 (1990); Dybvig, K. 1992. Gene Transfer In: Maniloff, J. (ed.) (ed.) Soc. Microbiol. Washington, D.C., pp.355-362)). M. genitalium possesses a single circular chromosome (Colman, S. D. et al., Gene 87:91-96 (1990); Peterson, S. N. et al., J. Bacteriol. 175:7918-7930 (1993)). M. genitalium’s genome has been hindered by a lack of auxotrophic mutations, and by a lack of a genetic exchange system that prevents reverse genetic approaches. The sequencing of M.genitalium’s genome will help us better understand how M.genitalium can cause or promote various invasive diseases or immunodeficiency disorders, and how to best combat mycoplasma infections.
Nucleic Acids Res. 16(13):6015-6025 (1988); Neimark, H. C. et.al., Nucleic Acids Res. 16(13):6015-6025 (1988); Neimark, H. C. et al., Nucleic Acids Res. The 18(18), 5443-5448, (1990)) showed that the genome sizes of mycoplasmas are very diverse. Southern blots hybridization of M. genitalium digested DNAs compared with the well-known pathogen M. pneumoniae showed low homology values, approximately 6-8 percent (Yogev D. and colleagues, International Journal of Microbiology, 1990). J. Syst. Bacteriol. 36(3):426-430 (1986)). There have been high homologies reported between the adhesin gene of M. pneumoniae and M. genitalium (Dallo S. F. 6:69-73 (1989)). Initial studies comparing the genomes of M. genitalium and the well-known M. pneumoniae species revealed that both species had three (3) rRNA gene clusters in a 5 kb chromosomal segment. They also formed a single operon, organized in the classical procaryotic manner, although there are differences between the restriction sites. J. Bacteriol. 36(3):426-430 (1986)).
Restriction Enzyme Mapping of M. Genitalium” indicates that the genome measures approximately 600 kb. Su, C. J. and colleagues, J. Bacteriol, have also mapped several genes, including the MgPa gene, which encodes the MgPa adhesion protein. 172:4705-4707 (1990); Colman, S. D. et al., Mol. Microbiol. 4(4):683-687 (1990)). The restriction map for the M. genitalium genome has been cloned as well in a library consisting of 20 cosmids overlapping each other and one clone (Lucier, T. S. et al., Gene 150:27-34 (1994)). clone (Lucier, T. S. et al., Gene 150:27-34 (1994)).
Nucleic Acids Review, 19:6027-6031 (1991) 19:6027-6031 (1991)). Using random sequencing on 508 nonidentical random clones, subsequent work has enabled sequence information to have been compiled for about seventeen percent (17%) of the M.genitalium genome. 175:7918-7930 (1993)). The sequence information shows that M. genitalium’s genome is small, but contains many genes involved in metabolic processes. The genome encodes approximately 390 protein, showing that M.genitalium is very efficient with its limited DNA. 175:7918-7930 (1993)).
Several studies were undertaken to sequence and characterize the individual genes found in M.genitalium. The medical importance of M.genitalium has helped direct attention to genes that determine the level of infection and virulence of the organism. The deduced amino-acid sequence of the MgPa adheresin gene (i.e. the gene that codes the surface cytadhesion proteins of M.genitalium) indicates that the complete MgPa gene contains 4,335 coding nucleotides for a protein 159,668 Da. (Dallo, S. F. et al., Infect. Immun. 57(4):1059-1065 (1989)). In addition, nucleotide sequences of the M.genitalium MgPa adheresin gene revealed specific codon orders for this important gene. (Inamine, J. M. and colleagues, Gene 82, 259-267, 1989)). MgPa adhesin also exhibited restriction fragment polymorphism, according to a study by Dallo, S. F. and colleagues in Microbial Path. 10:475-480 (1991)). Nucleotide homology to the well-known highly conserved procaryotic origin-of-replication gene (gyrA) was noted for M. genitalium (Bailey, C. C. et al., J. Bacteriol. 176:5814-5819 (1994)). The highly conserved procaryotic extension factor, Tu encoded by the Tuf gene, was noted and sequenced in M. genitalium. It contained an open reading frame that encoded a protein with approximately 393 amino acid (Loechel S. et.al., Nucleic acids Res. 17(23):10127 (1989)). It has been found that the tuf (M. genitalium) gene uses a different signal than the Shine-Delgamo sequence (ribosomal site binding site), preceding its initiation codon. (Loechel S. et.al., Nucleic acids Res. 19:6905-6911 (1991)).
The present invention is based upon the sequencing of the Mycoplasma genome. The primary nucleotide sequencing which was generated can be found in SEQID NO:1.
The present invention is a contiguous string of primary sequence information corresponding to the nucleotide pattern depicted in SEQ ID NO:1. In one embodiment, the present invention is presented as a string of primary sequence data corresponding to SEQ ID No:1 nucleotide.
The present invention also provides nucleotide sequencings that are at least 99.9% similar to the sequence of SEQ ID No:1.
To facilitate its use, “The nucleotide sequencing of SEQID NO:1, a fragment thereof or a nucleotide which is at most 99.9% identical with the sequence of SEQID NO:1 may be provided on a variety of media. The sequences of the invention can be recorded on computer-readable media in one embodiment of this invention. These media include, but are not limited to, magnetic storage media like floppy disks, hard disk storage medium and magnetic tape; optical media such CD-ROM; electronic storage media such RAM and ROM; hybrids of these media such as magnetic/optical media.
The present invention also provides systems, namely computer-based systems that contain the sequence data described herein stored in a storage medium. These systems are designed to identify commercially significant fragments of Mycoplasma genome.
Another embodiment of the invention relates to fragments isolated from the Mycoplasma genome. “The fragments of Mycoplasma genitalium’s genome that are the subject of this invention include but are not restricted to fragments that encode peptides. These fragments will be called open reading frames, or ORFs. They may also be fragments modulating the expression of a operably linked ORF.
Each ORF fragment of the Mycoplasma Genitalium genome revealed in Tables 1(a),1(c)and 2, and the EMF discovered 5? The ORF can be used as polynucleotide primers in a variety of ways. These sequences are useful as diagnostic probes and diagnostic amplification primers to detect the presence of microbes in a sample. They can also be used for the production commercially valuable pharmaceutical agents or to control gene expression.
The present invention also includes recombinant constructions that contain one or more fragments from the Mycoplasma genome. The recombinant constructions of the invention include vectors such as a viral or plasmid vector into which a Mycoplasma gene fragment has been inserted.
The present invention also provides host cells that contain any of the fragments of Mycoplasma genome isolated in the present invention. The host cells may be higher eukaryotic hosts such as mammalian cells, lower eukaryotic hosts such as yeast cells, or procaryotics such as bacteria.
Click here to view the patent on Google Patents.
Leave a Reply