Invented by David S. Ojala, Jorge Santiago-Ortiz, Oscar Westesson, David V. Schaffer, Ian H. Holmes, John Weinstein, University of California
Adenoassociated viruses (AAVs) have gained significant attention in recent years due to their potential as gene therapy vectors. AAVs are small, non-pathogenic viruses that have been extensively studied and engineered to deliver therapeutic genes to target cells. This has led to a growing market for AAV variants and the development of innovative methods for their use.
The global gene therapy market has been expanding rapidly, with AAV-based therapies being one of the key drivers. AAVs have several advantages over other viral vectors, such as their ability to infect both dividing and non-dividing cells, low immunogenicity, and long-term gene expression. These characteristics make AAVs an attractive choice for gene therapy applications, including the treatment of genetic disorders, cancer, and other diseases.
The market for AAV variants is driven by the need for improved vectors with enhanced transduction efficiency, tissue specificity, and safety profiles. Researchers and biotech companies are constantly working on developing novel AAV serotypes and capsid variants to overcome the limitations of existing vectors. For example, AAV9 has shown great promise in crossing the blood-brain barrier, making it a potential candidate for treating neurological disorders. Similarly, AAVrh10 has demonstrated efficient transduction of lung cells, making it a valuable tool for respiratory gene therapy.
In addition to serotype engineering, several methods have been developed to improve the performance of AAV vectors. One such method is directed evolution, which involves the generation of large libraries of AAV variants and their screening for desired properties. This approach has led to the identification of AAV variants with improved transduction efficiency, altered tissue tropism, and reduced immunogenicity.
Another method for enhancing AAV vector performance is the use of hybrid vectors. These vectors combine the best features of different AAV serotypes to create chimeric vectors with improved transduction efficiency and tissue specificity. For example, the hybrid vector AAV-DJ has been shown to transduce a wide range of tissues, making it a valuable tool for gene therapy applications.
Furthermore, advancements in manufacturing technologies have also contributed to the growth of the AAV variant market. The development of scalable and cost-effective production methods, such as transient transfection and stable cell lines, has made it easier to produce large quantities of AAV vectors for preclinical and clinical studies. This has not only accelerated the development of AAV-based therapies but has also made them more accessible to a wider range of researchers and companies.
In conclusion, the market for AAV variants is expanding rapidly, driven by the increasing demand for improved gene therapy vectors. The development of novel AAV serotypes, capsid variants, and hybrid vectors, along with advancements in manufacturing technologies, has opened up new possibilities for gene therapy applications. As research in this field continues to progress, we can expect to see further innovations in AAV vector design and methods for their use, ultimately leading to more effective and safer gene therapies for various diseases.
The University of California invention works as follows
The present disclosure provides recombinant AAV virions that contain a variant AAV protein, such as an AAV protein derived from a ancestral AAV protein amino acid sequence. The rAAV virus of the present disclosure is capable of infecting a cell more effectively. The present disclosure provides also methods for delivering a genetic product to a cell target in an individual through the administration of an rAAV. The present disclosure provides also methods for generating rAAV viruses that have a variant AAV protein derived an ancestral AAV protein amino acid sequence.
Background for Adenoassociated virus variants, and methods for their use
The present disclosure provides recombinant AAV virions that contain a variant AAV protein, such as an AAV protein derived from a ancestral AAV protein amino acid sequence. The rAAV virus of the present disclosure is capable of infecting a cell more effectively. The present disclosure provides also methods for delivering a genetic product to a cell target in an individual through the administration of an rAAV. The present disclosure provides also methods for generating rAAV viruses that have a variant AAV protein derived an ancestral AAV protein amino acid sequence.
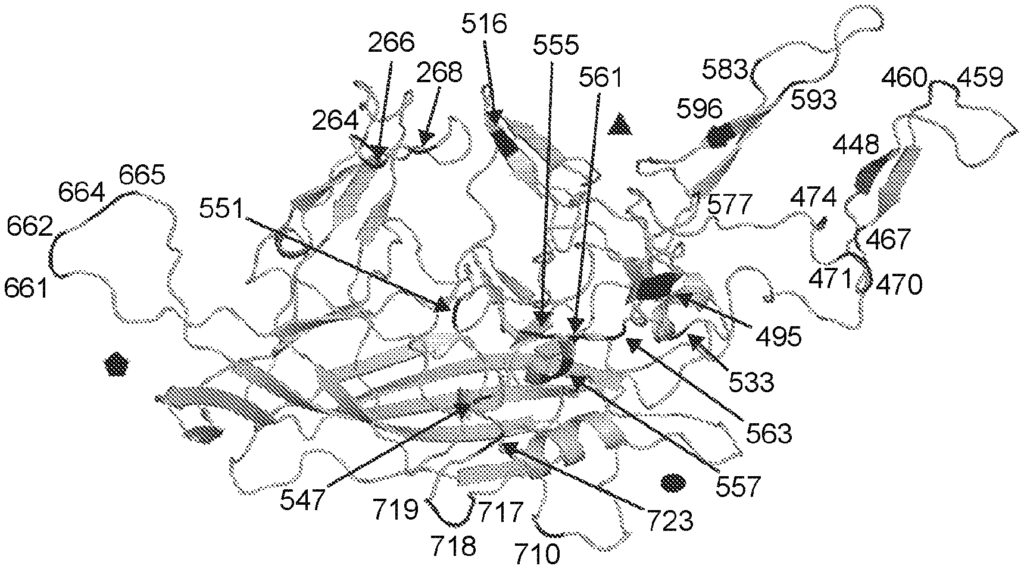
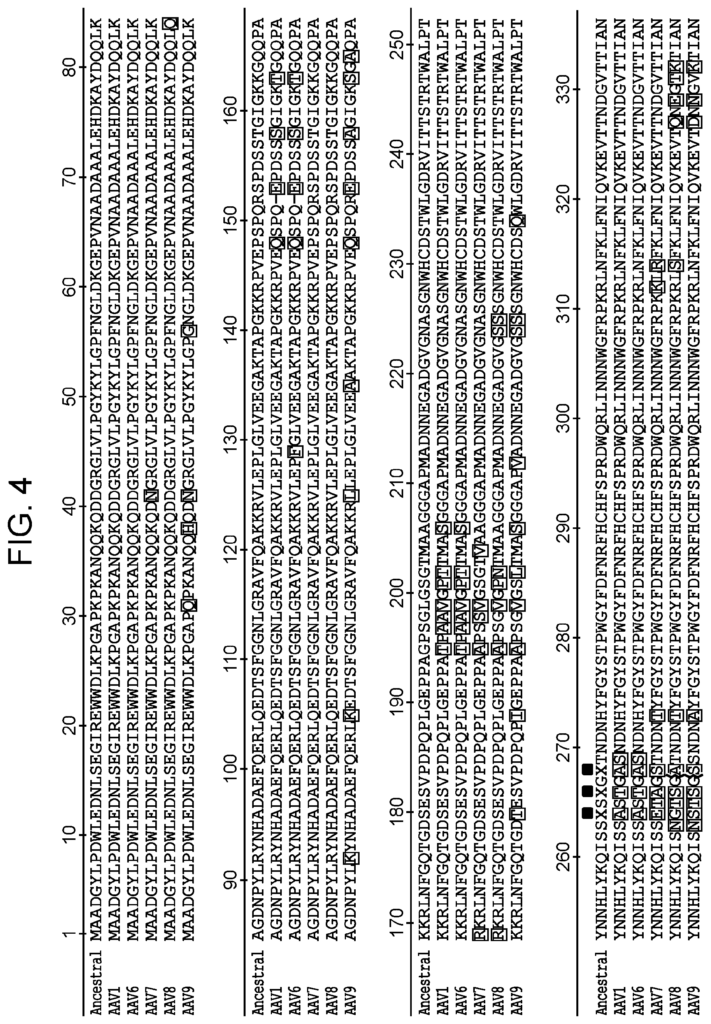
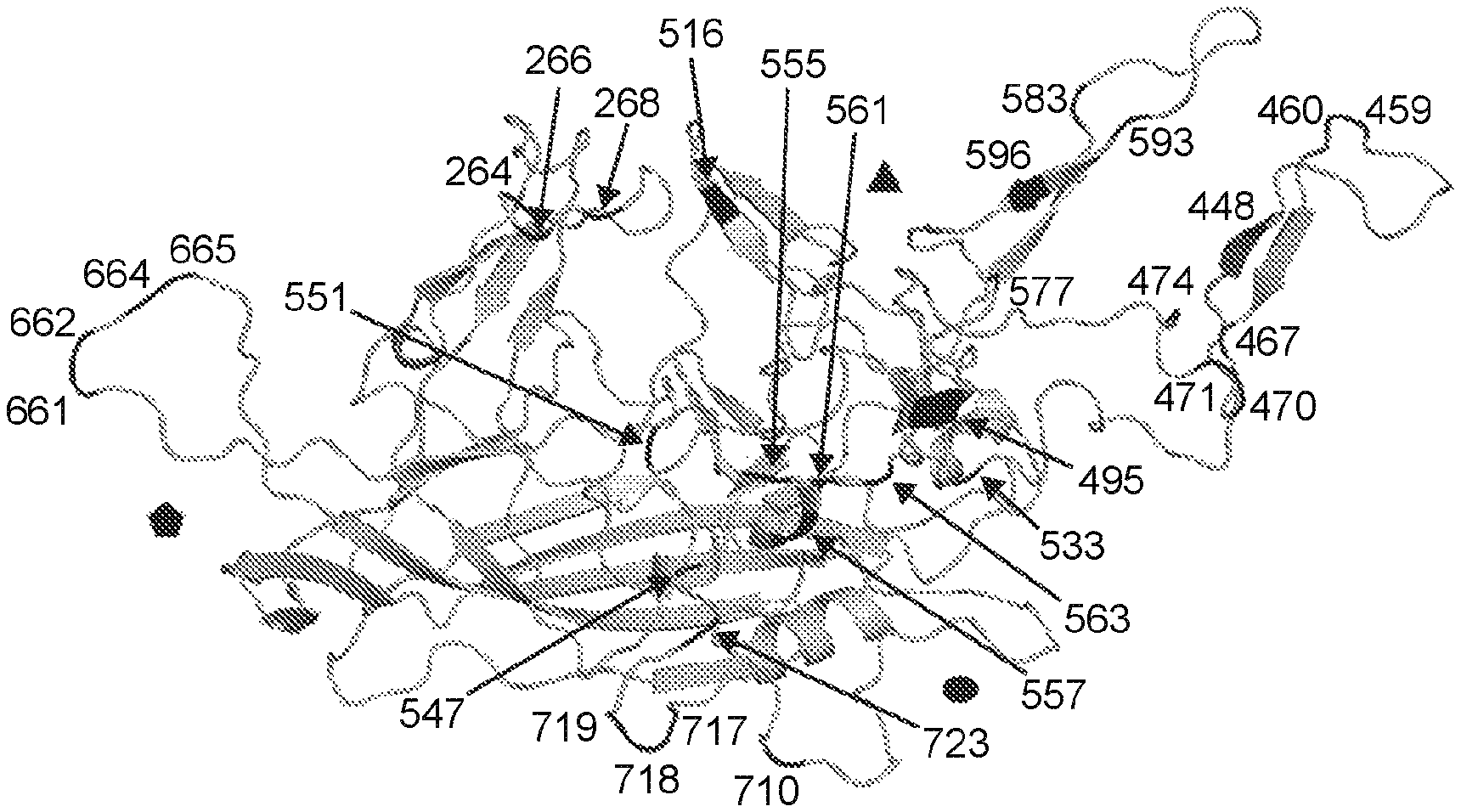
The present disclosure includes: “Aspects include an rAAV virus containing: a.) a variant AAV protein, which contains an amino acid-sequence having at least a 95% identity to that of SEQ ID No: 16; wherein, the amino acids are at positions 264; 448; 459; 470; 495; 533; 547; 555; 557; 561, 563; 593; 596; 661; 662; 664 In certain embodiments the variant AAV protein includes an amino-acid sequence that has at least 95% sequence identity with the sequence of SEQ ID No: 13. In certain embodiments the variant AAV protein contains an amino acid that has at least 95% sequence identity with the sequence in SEQ NO: 14. In certain embodiments the variant AAV protein capsid includes an amino sequence that has at least 95% sequence identity with the sequence described in SEQ NO: 14.
In some embodiments, variant AAV proteins, when present in rAAV virions, confer increased infectivity to a target cell. The target cell can be a muscle or glial cells in some cases. In some embodiments, rAAV has at least a 5-fold increase in infectivity compared to an AAV virus containing a wild-type AAV serotype capid protein.
In some embodiments, a variant AAV protein confers altered dependencies on target cell receptors compared to a wild-type AAV serotype protein. In some embodiments the rAAV virus has a reduced dependence on sialic acid or heparin-sulfate prooglycans to infectivity as compared to AAV virion containing a wild type AAV serotype capsid proteins.
In any embodiment, the polypeptide may be troponin, laminin, collagen, lamin, selenoprotein N, protein-O-mannosyltransferase, fukutin, acetylglucosaminyltransferase-like 1A (also known as LARGE1), O linked mannose?1,2-N acetylglucosaminyltransferase or isoprenoid syntha In any embodiment, the polypeptide may be a troponin, laminin, collagen, lamin, selenoprotein N, protein-O-mannosyltransferase, fukutin, acetylglucosaminyltransferase-like 1A (also known as LARGE1), O-linked mannose ?1,2-N-acetylglucosaminyl-transferase, or isoprenoid synthase domain-containing protein. In any of the embodiments discussed herein, the secreted polypeptide may be lipoprotein lipase, factor IX, ?1-antitrypsin, follistatin, soluble myostatin receptor, apelin, glucagon-like peptide 1, insulin-like growth factor 1, alpha-galactosidase, iduronidase, iduronate-2-sulfatase, alpha-glucosidase, and N-acetylgalactosamine 4-sulfatase.
In any embodiment, the product may be a genome-editing gene product. This could include zinc finger nucleases and transcription activator like effector nucleases.
In any embodiment, the nucleic-acid gene product can be an interfering nucleic-acid, a ribozyme or antisense nucleic acids, or even an aptamer.
Also, provided herein are pharmaceutical compositions containing an rAAV virus according to any embodiment described above or below, and a pharmaceutically accepted carrier, diluent or excipient.
Also, provided herein is an embodiment of a method for delivering a genetic product to a cell target in an individual. The method comprises administering an rAAV virus to the individual according to any embodiment described above or below. In some embodiments the target cell can be a muscle or a glial cells.
Other aspects of the disclosure include an isolate nucleic acid that contains a nucleotide-sequence encoding a variant AAV capid protein. The variant AAV capid proteins includes an amino acid series having at least 95% sequence identity to SEQ ID No: 16 where the amino acids are A or A.S, N.A, S.E, Q.T.D.M, S.Q.T., A.V, S.S. and S.T. In certain embodiments, the variant AAV capsid proteins, when present in a virion, provide for an increased infectivity for a muscle or glial cells compared to an AAV virus comprising the wild type AAV cap protein.
Also, provided herein are an isolated, genetically-modified host cell containing nucleic acids of any embodiment shown above or below.
The variant AAV protein is also provided, and includes an amino-acid sequence that has at least 95% identity to SEQ ID No: 16; wherein, the amino-acid sequence at positions 264, 448, 458, 459, 470, 495, 533, 551, 561, 563, 593, 596, 661, 662, 664, 718, and 723 is A, A, N, S, S, In certain embodiments, a variant AAV capsid proteins confers greater infectivity to a muscle cell or glial cells than an AAV virus comprising the wild type AAV cap protein.
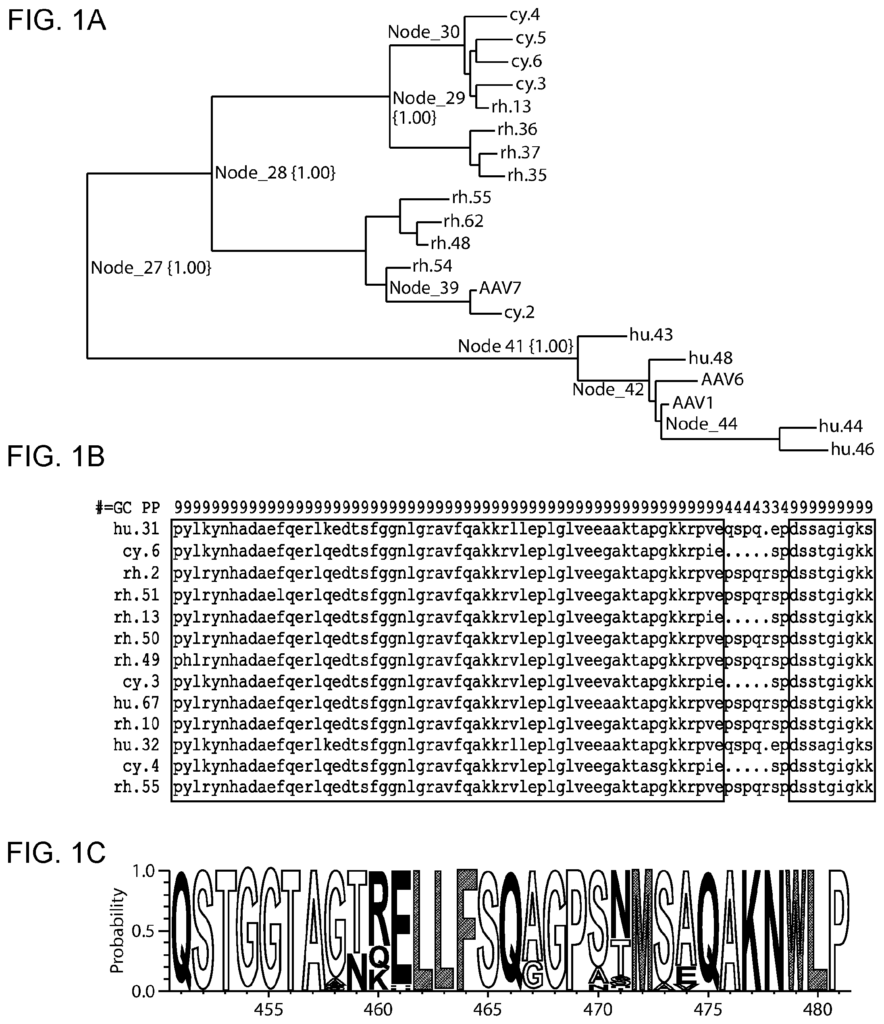
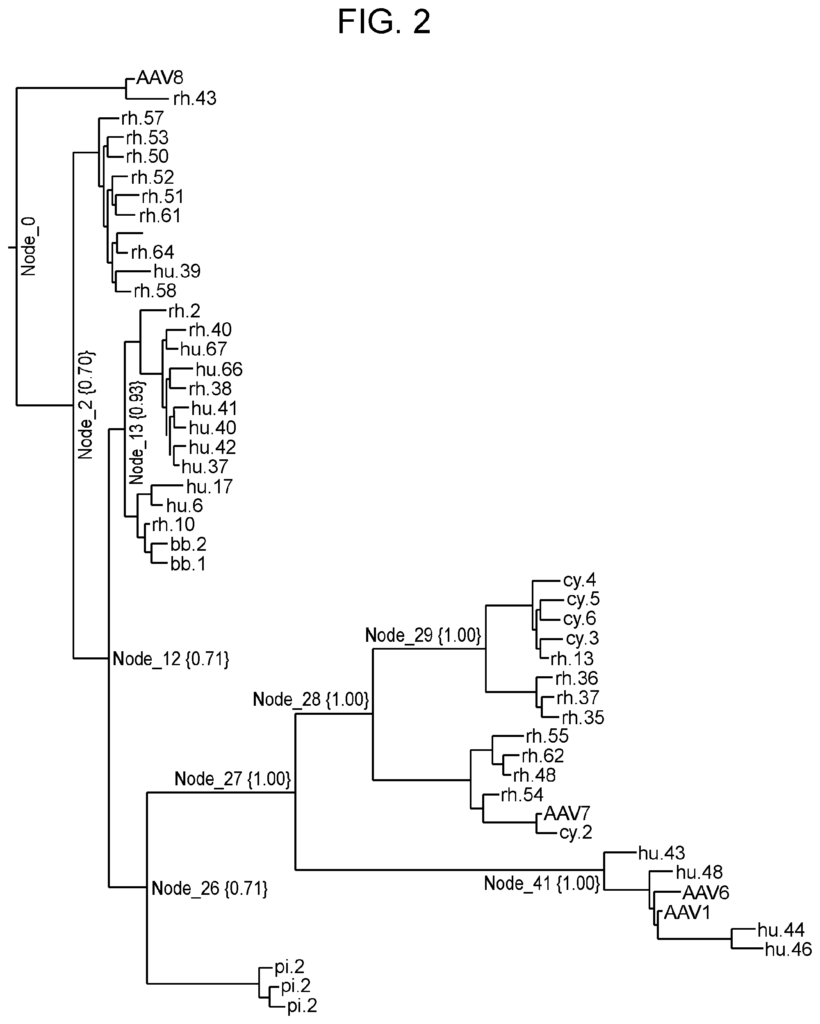
The present disclosure also includes a method for generating rAAV viruses having variant AAV proteins. This method involves subjecting a library of initial rAAV virus to a selection process in target cells. Each rAAV virus in the library contains an initial AAV protein with an AAV protein amino-acid sequence. In some embodiments the method also includes determining an ancestral AAV protein amino-acid sequence by i.) reconstructing a tree of a number of wild type AAV protein amino-acid sequences; ii.) selecting a branch of the tree and iii.) determining the amino acid at that branch. In some embodiments the method also includes estimating confidence values at each node in the phylogenetic trees, and selecting a node based on that estimated confidence value. In some cases, the AAV capsid has an amino-acid sequence that is at least 94% the same as the sequence in SEQ NO: 7.
In any embodiment, the method can include subjecting the second rAAV virus library to a second selection round. In some embodiments the second round is done in the same type of target cell as was used in the initial round. In some embodiments the second round has a higher level of stringency than the first round.
In any embodiment, target cells can be muscle cells or epithelial, skin, or glial cell types. In some cases the target cells may be human embryonic renal cells.
In some embodiments the subjecting step involves a) superinfecting infected target cells with a virus helper, and harvesting released rAAV viruses from superinfected cell.
The method can be characterized by the following: “In any embodiment the method may include isolating an rAAV virus containing variant AAV proteins, which has enhanced infectivity or tropism compared to AAV viruses containing wild type AAV serotypes capsid proteins.
Kits that contain the rAAV viruses or libraries of rAAV virus are also provided. These kits can be used to practice the methods.
Recombinant Adeno-Associated Virus (rAAV), virions, comprising a variant AAV proteins, e.g. an AAV protein derived an ancestral AAV protein amino acid sequence are provided. In certain embodiments, the rAAV viruses comprising the variant AAV proteins exhibit greater infectivity towards target cells such as muscle and glial cell. Herein, methods are also provided for delivering a genetic product to a cell target in an individual through the administration of rAAV viruses with the variant capsid proteins. The present disclosure also provides methods for generating rAAV viruses that have a variant AAV proteins derived from an AAV protein amino acid sequence.
Recombinant Adenoassociated Virus Virions

The present disclosure provides rAAV viruses comprising a variant AAV proteins, e.g. an AAV protein derived from ancestor AAV protein amino acid sequence.
Click here to view the patent on Google Patents.
Leave a Reply