Invented by Charles David Pauza, Haishan Li, Tyler Lahusen, Mei-Ling Liou, American Gene Technologies International Inc
The activation of gamma-delta cells is essential for their effector functions, including cytokine production, cytotoxicity, and antigen presentation. However, compared to alpha-beta (??) T cells, the mechanisms of gamma-delta cell activation are not well understood. Therefore, there is a significant need for methods that can specifically and efficiently activate these cells to harness their potential in immunotherapy.
One approach to activating gamma-delta cells is through the use of synthetic antigens or antibodies that specifically bind to gamma-delta T cell receptors (TCRs). These antigens or antibodies can stimulate the TCR signaling pathway, leading to gamma-delta cell activation. Several companies are developing and commercializing such synthetic antigens or antibodies for research and therapeutic purposes.
Another promising method of activating gamma-delta cells is through the use of cytokines or chemokines that can selectively recruit and activate these cells. For example, interleukin-2 (IL-2) and interleukin-15 (IL-15) have been shown to enhance the proliferation and cytotoxicity of gamma-delta cells. Companies are actively exploring the use of these cytokines, either alone or in combination with other immunomodulatory agents, to activate gamma-delta cells for cancer immunotherapy.
Furthermore, researchers are investigating the potential of small molecules or drugs that can modulate the activation of gamma-delta cells. These molecules can target specific signaling pathways or receptors involved in gamma-delta cell activation, thereby enhancing their effector functions. The development of such small molecules holds great promise for the treatment of various diseases, including cancer and infectious diseases.
The market for methods of activation of gamma-delta cells is driven by the increasing understanding of the unique functions and therapeutic potential of these cells. The growing prevalence of cancer and infectious diseases, coupled with the limitations of current treatment options, has created a demand for innovative immunotherapies. Gamma-delta cell-based therapies offer a novel approach to harnessing the immune system’s power to fight diseases.
Moreover, the market is also fueled by the advancements in biotechnology and genomics, which have enabled the identification and characterization of gamma-delta cell subsets and their specific activation mechanisms. These technological advancements have paved the way for the development of targeted therapies that can selectively activate gamma-delta cells, minimizing off-target effects and improving treatment outcomes.
In conclusion, the market for methods of activation of gamma-delta cells is a rapidly expanding field with significant potential in the biotechnology and pharmaceutical industries. The development of synthetic antigens, cytokines, small molecules, and other immunomodulatory agents holds promise for harnessing the unique functions of gamma-delta cells in immunotherapy. As research continues to unravel the complexities of gamma-delta cell biology, we can expect to see further advancements in this field, leading to the development of novel and more effective therapies for various diseases.
The American Gene Technologies International Inc invention works as follows
The present invention is a method and composition for gene therapy and immunetherapy that activates gamma-delta T-cells. It can be used to treat various cancers and infectious disease.
Background for Methods of activation of Gamma-delta cells
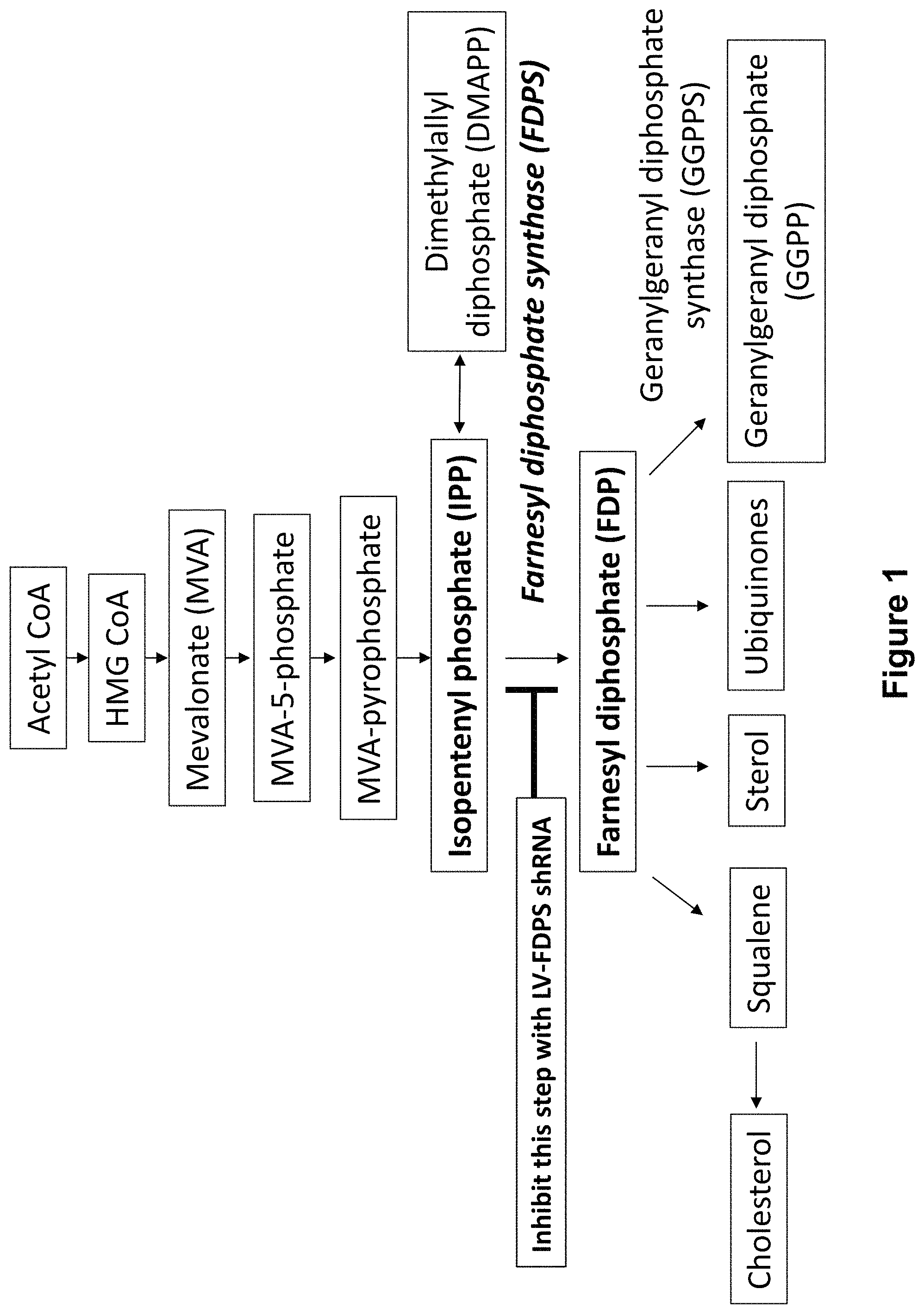
The T cell receptor is used to distinguish human T cells. Major populations, such as CD4+ and CD8+, express receptors composed of alpha- and beta-chains. A smaller subset of T cells expresses a receptor composed of gamma-delta chains. Gamma delta (?GD?) T cells are 3-10% of the circulating lymphocytes. The V?2+ subset is 75% of all GD T-cells in blood. V?2+ T cells can recognize non-peptide antigens without the need for antigen presentation via major histocompatibility (?MHC?) complexes. or human leukocyte antigen (?HLA?). Most V?2+ cells express a V9 chain, and they are stimulated when exposed to compounds with 5-carbon pyrophosphates that are intermediates of the mevalonate or non-mevalonate pathways for sterol/isoprenoid syntheses. “The response to 5-carbon isopentenyl-pyrophosphate is universal in healthy humans.
The epithelial mucosa, and skin, are common sites for V?+1 T cells.
In general, GD cells are capable of killing pathogens and tumor cells. Their unique T cell receptors (?TCRs?) stimulate them. The unique T cell receptor (?TCRs?) composed of two glycoprotein chain,? The? and? chains, which are composed of two glycoprotein chains, enhance cellular cytotoxicity and cytokine release, as well as other effector functions. The TCRs on GD T-cells are unique and they occur at high clonal frequency. This allows rapid responses to pathogens and tumors.
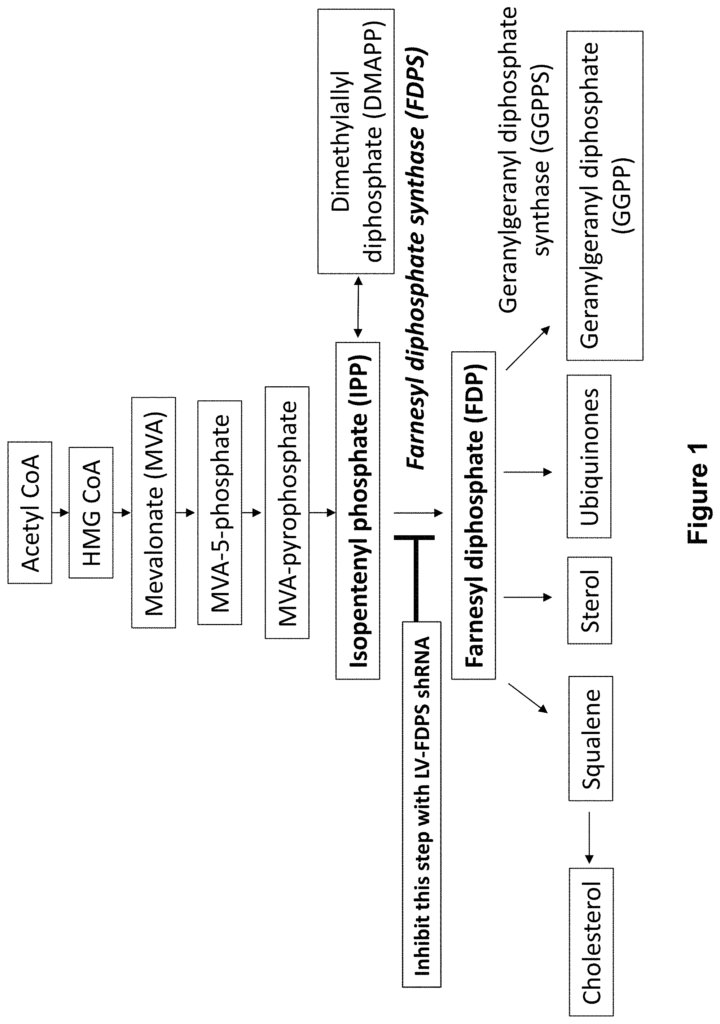
ABPs can also be used to stimulate GD cells. It may be that when FDPS in myeloid cell is inhibited, IPP starts to accumulate. Geranylgeranyl pyrophosphate(?GGPP?) also increases. The downstream product of FDPS, geranylgeranyl pyrophosphate (?GGPP), which suppresses the activation of inflammasome pathways is reduced. “The reduction of GGPP removes a caspase-dependent pathway inhibitor and allows the secretion mature cytokines such as interleukin-18 and interleukin-beta, which are especially important for gamma-delta T cell activation.
When FDPS is blocked the IPP increases and GGPP decreases, which combine to activate V2+ T-cells. V?2+ T cells that are activated by ABPs or IPPs will multiply rapidly, release a variety of cytokines and other chemokines and function as cytotoxic destroyers of tumor cells and cells infected with microorganisms.
However ABPs have been associated with osteonecrosis and inflammation, in addition to having a poor bioavailability because of their chemistry. IPP is also difficult to synthesize and has a short half-life. Both compounds must be administered systemically to an individual. Both ABPs and IPPs, in general, are not very effective for therapeutic purposes.
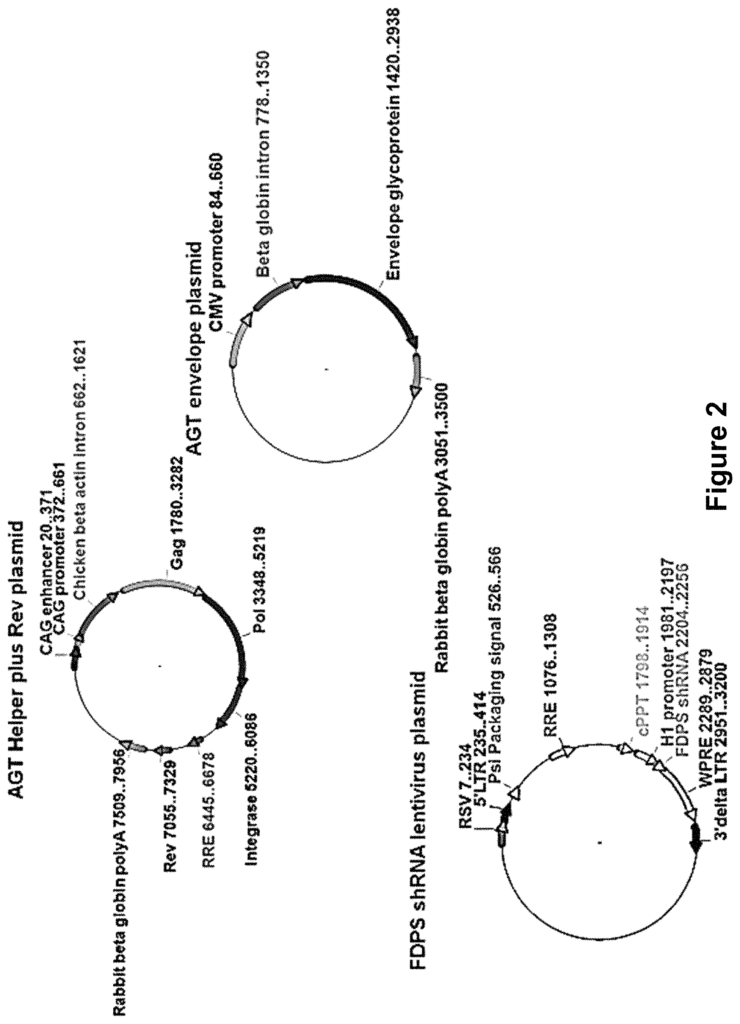
In one aspect, a technique for activating a GD-T cell is described. Infecting a target with a viral system that encodes a genetic element is part of the method. In some embodiments, at least one of the genetic elements is a small RNA that inhibits the production of an enzyme in the mevalonate path. In embodiments, FDPS is the enzyme. In some embodiments, if the enzyme in the target cells is inhibited, the cell will activate the GD T-cell. In some embodiments, the cell being targeted is either a cancerous cell or one that has been infected by an infectious agent. In a preferred embodiment the activation results in the GD cell killing the cancerous cell or cell that has been infected by an infectious agent. In some embodiments, at least one genetic element encoded includes a shRNA or microRNA. In other embodiments, an aminobisphosphonate is also applied to the target cell. In some embodiments, zoledronic is the aminobisphosphonate.
In another aspect, there is provided a method for treating cancer in a patient. The method includes administering to the subject a therapeutically-effective amount of a viral delivery system that encodes at least one genetic element. In some embodiments, at least one of the genetic elements is a small-RNA capable of blocking production of an enzyme in the mevalonate path. In other embodiments, if the enzyme in a cancerous cell is inhibited when a GD-T cell is present, the cancerous cell will activate the GD-T cell to treat the cancer. In some embodiments, FDPS is the enzyme. In some embodiments, at least one genetic element encoded includes a shRNA or microRNA. In other embodiments, an aminobisphosphonate is also applied to the target cell. In some embodiments, zoledronic is the aminobisphosphonate.
In another aspect, the method is to treat an infectious disease on a subject. The method includes administering to the subject a therapeutically-effective amount of a viral delivery system that encodes at least one genetic element. In some embodiments, at least one of the genetic elements is a small-RNA capable of blocking the production of an enzyme in the mevalonate path. In other embodiments, when an enzyme is inhibited by a cell infected with a pathogen in the presence a GD cell, the cell infected activates the GD cell to treat the cell infected and the infectious disease. In some embodiments, FDPS is the enzyme. In some embodiments, at least one genetic element encoded includes a microRNA (or shRNA). In other embodiments, an aminobisphosphonate is also applied to the target cell. In some embodiments, zoledronic is the aminobisphosphonate.
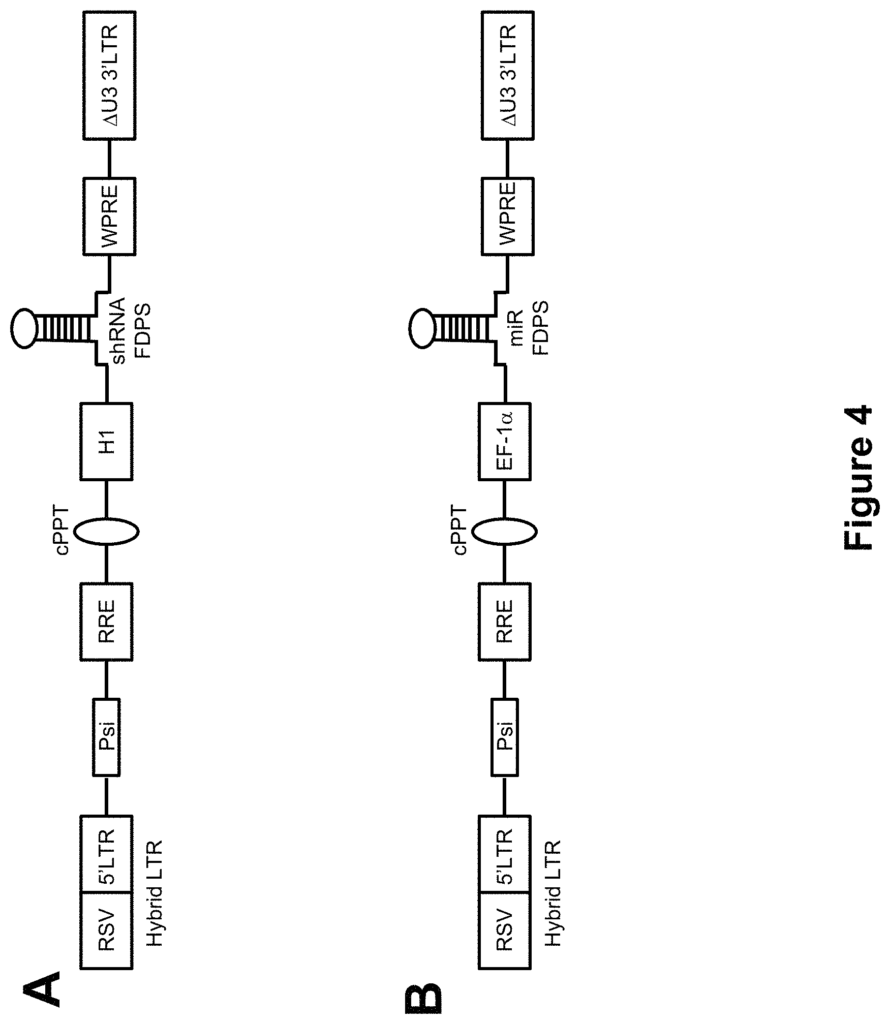
In another aspect, the at least one encoded genetic element includes a shRNA having at least 80%, or at least 85%, or at least 90%, or at least 95% percent identity with GTCCTGGAGTACAATGCCATTCTCGAGAATGGCATTGTACTCCAGGACTTTTT (SEQ ID NO: 1); GCAGGATTTCGTTCAGCACTTCTCGAGAAGTGCTGAACGAA ATCCTGCTTTTT (SEQ ID NO: 2); GCCATGTACATGGCAGGAATTCTCGAGAA TTCCTGCCATGTACATGGCTTTTT (SEQ ID NO: 3); or GCAGAAGGAGGCTGA GAAAGTCTCGAGACTTTCTCAGCCTCCTTCTGCTTTTT (SEQ ID NO: 4). In a preferred embodiment, the shRNA includes GTCCTGGAGTACAATGCCATTCTCGAG AATGGCATTGTACTCCAGGACTTTTT (SEQ ID NO: 1); GCAGGATTTCGTTCA GCACTTCTCGAGAAGTGCTGAACGAAATCCTGCTTTTT (SEQ ID NO: 2); GCCA TGTACATGGCAGGAATTCTCGAGAATTCCTGCCATGTACATGGCTTTTT (SEQ ID NO: 3); or GCAGAAGGAGGCTGAGAAAGTCTCGAGACTTTCTCAGCCTCCTT CTGCTTTTT (SEQ ID NO: 4).
In another aspect, the at least one encoded genetic element includes a microRNA having at least 80%, or at least 85%, or at least 90%, or at least 95% percent identity with AGGTATATTGCTGTTGACAGTGAGCGACACTTTCTCAGCCTCCTTCTGCGTGAAG CCACAGATGGCAGAAGGAGGCTGAGAAAGTGCTGCCTACTGCCTCGGACTTCAA GGGGCT (SEQ ID NO: 5); AAGGTATATTGCTGTTGACAGTGAGCGACACT TTCTCAGCCTCCTTCTGCGTGAAGCCACAGATGGCAGAAGGGCTGAGAAAGTGCT GCCTACTGCCTCGGACTTCAAGGGGCT (SEQ ID NO: 6); TGCTGTTGACAGTG AGCGACTTTCTCAGCCTCCTTCTGCGTGAAGCCACAGATGGCAGAAGGAGGCTG AGAAAGTTGCCTACTGCCTCGGA (SEQ ID NO: 7); CCTGGAGGCTTGCTGAAG GCTGTATGCTGACTTTCTCAGCCTCCTTCTGCTTTTGGCCACTGACTGAGCAGAAG GGCTGAGAAAGTCAGGACACAAGGCCTGTTACTAGCACTCA (SEQ ID NO: 8); CATCTCCATGGCTGTACCACCTTGTCGGGACTTTCTCAGCCTCCTTCTGCCTGTTG AATCTCATGGCAGAAGGAGGCGAGAAAGTCTGACATTTTGGTATCTTTCATCTGA CCA (SEQ ID NO: 9); or GGGCCTGGCTCGAGCAGGGGGCGAGGGATACTTTCT CAGCCTCCTTCTGCTGGTCCCCTCCCCGCAGAAGGAGGCTGAGAAAGTCCTTCCC TCCCAATGACCGCGTCTTCGTCG (SEQ ID NO: 10). In a preferred embodiment, the microRNA includes AAGGTATATTGCTGTTGACAGTGAGCGACACTTTCTCAGCCT CCTTCTGCGTGAAGCCACAGATGGCAGAAGGAGGCTGAGAAAGTGCTGCCTACT GCCTCGGACTTCAAGGGGCT (SEQ ID NO: 5); AAGGTATATTGCTGTTGACAGT GAGCGACACTTTCTCAGCCTCCTTCTGCGTGAAGCCACAGATGGCAGAAGGGCTG AGAAAGTGCTGCCTACTGCCTCGGACTTCAAGGGGCT (SEQ ID NO: 6); TGCTG TTGACAGTGAGCGACTTTCTCAGCCTCCTTCTGCGTGAAGCCACAGATGGCAGAA GGAGGCTGAGAAAGTTGCCTACTGCCTCGGA (SEQ ID NO: 7); CCTGGAGGCT TGCTGAAGGCTGTATGCTGACTTTCTCAGCCTCCTTCTGCTTTTGGCCACTGACTG AGCAGAAGGGCTGAGAAAGTCAGGACACAAGGCCTGTTACTAGCACTCA (SEQ ID NO: 8); CATCTCCATGGCTGTACCACCTTGTCGGGACTTTCTCAGCCTCCTT CTGCCTGTTGAATCTCATGGCAGAAGGAGGCGAGAAAGTCTGACATTTTGGTATC TTTCATCTGACCA (SEQ ID NO: 9); or GGGCCTGGCTCGAGCAGGGGGCGAGGG ATACTTTCTCAGCCTCCTTCTGCTGGTCCCCTCCCCGCAGAAGGAGGCTGAGAAA GTCCTTCCCTCCCAATGACCGCGTCTTCGTCG (SEQ ID NO: 10).
In another aspect is a viral vector containing at least one genetically encoded element. The at least encoded genetic elements includes a small-RNA capable of blocking production of an enzyme in the mevalonate path. In some embodiments, farnesyl dimphosphate synthase is the enzyme that participates in the mevalonate path. In some embodiments, at least one genetic element encoded includes a microRNA (or shRNA).
In another aspect, at least one encoded element includes a shRNA that has at least 80% identity, or even 85% or 90% or 95%. In a preferred embodiment the shRNA contains SEQ NO: 1, SEQ NO: 2, SEQ NO: 3, or SEQ NO: 4.
In another aspect, at least one genetic element encoded includes a microRNA that shares at least 80% or at most 85% or at minimum 90% or at the very least 95% of its identity with SEQ NO: 5, SEQ NO: 6, SEQ NO: 7, SEQ NO: 8, SEQ NO: 9, or SEQ NO: 10. In a preferred embodiment of the microRNA, SEQ NO: 5, SEQ NO: 6, SEQ NO: 7, SEQ NO: 8, SEQ NO: 9, or SEQ NO: 10.
In embodiments, a viral vector can be any vector capable of effectively transducing the small RNA to a target cell. In some embodiments, lentiviral is the viral vector. In other embodiments the viral vector may be an adenoassociated virus vector.
In another aspect the viral vector comprises a second genetic element encoded.” In some embodiments, at least one cytokine and/or chemokine is included in the second genetic component. In embodiments, at least one cytokine can be selected from a group including: IL-18 (or TNF-?) interferon?, IL-2 (or IL-15), IL-17 (or IL-12), IL-12, IL-15 or IL-17. In embodiments, at least one chemokine can be a CC chemokine (or a CXC chemokine). In other embodiments, at least one of the chemokines is RANTES.
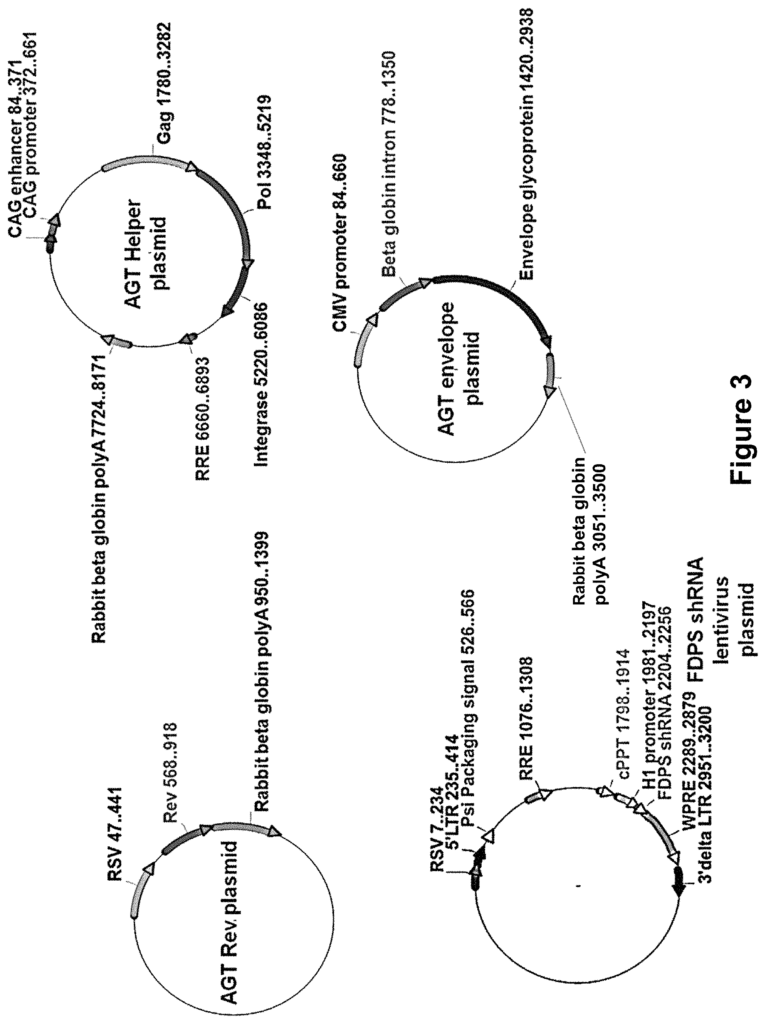
In another aspect, there is a lentiviral system that expresses a particle of lentiviral virus. The system comprises a lentiviral plasmid, at least a envelope plasmid expressing an optimized envelope protein for infecting cells, and at least a helper plasmid expressing gag genes, pol genes, and rev gene. The packaging cell produces a lentiviral particles when the lentiviral virus, the envelope plasmid and the helper plasmids are transfected. In some embodiments, a lentiviral particles is capable of infecting and inhibiting the enzyme in the mevalonate path within a target cell. In embodiments, FDPS is the enzyme that participates in the mevalonate path. In some embodiments, lentiviral system vectors include a first plasmid to express the gag, pol, and rev genes and a second plasmid to express the rev gene. In embodiments, envelope proteins are preferably optimized to infect a target cell. In some embodiments, the cancer cell is the target cell. In other embodiments the target cell may be a cell infected by an infectious agent.
Overview of Disclosure
The present disclosure is about gene therapy constructs, and their delivery to cells. This results in the suppression of Farnesyl Diphosphate Synase (FDPS). “The present disclosure relates to gene therapy constructs and delivery of the same to cells, resulting in suppression of Farnesyl diphosphate synthase (?FDPS? 1 . In some embodiments, microRNAs and short homology RNAs target FDPS to reduce the expression of this enzyme. Viral vectors can be lentiviral vectors or AAV vectors. Modulating the expression of FDPS can lead to an increase in IPP accumulation, which is a stimulator for GD T cells proliferation and differentiation. The constructs described herein can be used to activate GD cells and to treat cancer and infectious diseases.
Definitions and Interpretation
Click here to view the patent on Google Patents.
Leave a Reply