Invented by Larry J. Miller, Teleflex Life Sciences Ltd
The market for apparatus for monitoring, supporting and/or installing intraosseous devices is driven by several factors. Firstly, the increasing prevalence of chronic diseases, such as diabetes and cancer, is leading to a rise in the number of patients who require intravenous access. Secondly, the increasing incidence of trauma and accidents is also contributing to the growth of this market. Finally, the increasing awareness among healthcare professionals about the benefits of intraosseous access is also driving the growth of this market.
The apparatus for monitoring, supporting and/or installing intraosseous devices includes a range of products such as needles, catheters, drills, and monitoring systems. These products are designed to make the insertion of intraosseous devices easier and safer for healthcare professionals. They also help to monitor the patient’s condition during the procedure and ensure that the device is functioning properly.
The market for apparatus for monitoring, supporting and/or installing intraosseous devices is highly competitive, with several players operating in the market. Some of the key players in this market include Teleflex Medical, Cook Medical, Becton, Dickinson and Company, and PerSys Medical. These companies are investing heavily in research and development to develop innovative products that can improve patient outcomes and reduce the risk of complications during the insertion of intraosseous devices.
In conclusion, the market for apparatus for monitoring, supporting and/or installing intraosseous devices is growing rapidly, driven by several factors such as the increasing prevalence of chronic diseases, trauma, and accidents. The key players in this market are investing heavily in research and development to develop innovative products that can improve patient outcomes and reduce the risk of complications during the insertion of intraosseous devices. As the demand for intraosseous access continues to grow, the market for apparatus for monitoring, supporting and/or installing intraosseous devices is expected to continue to expand in the coming years.
The Teleflex Life Sciences Ltd invention works as follows
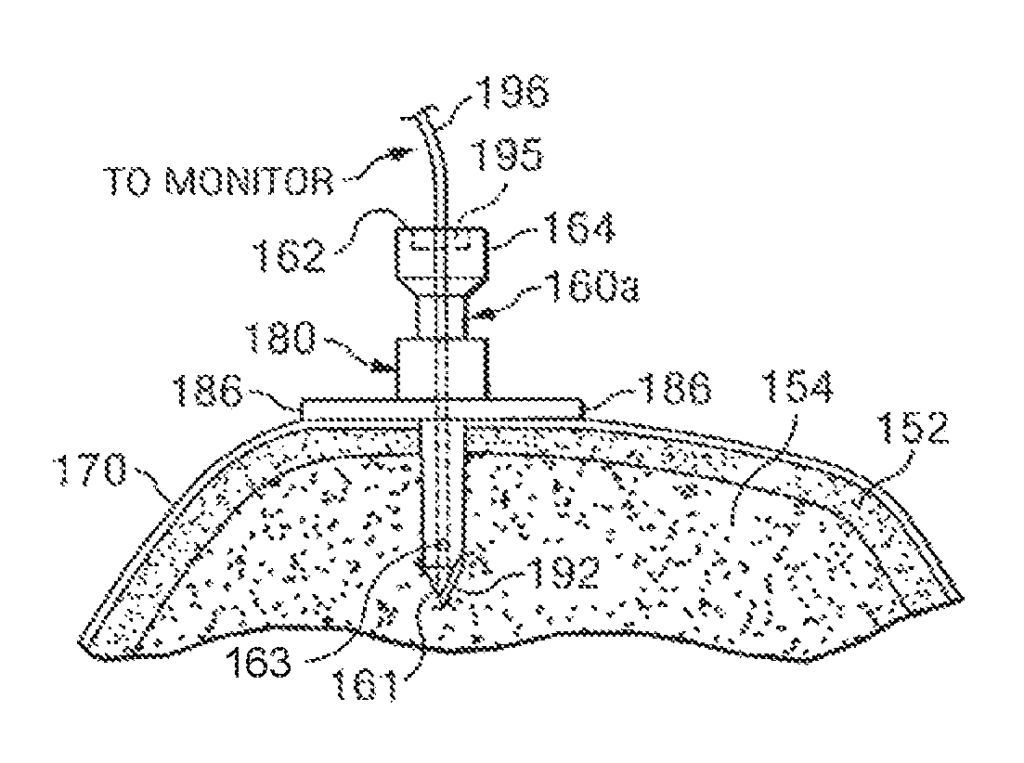
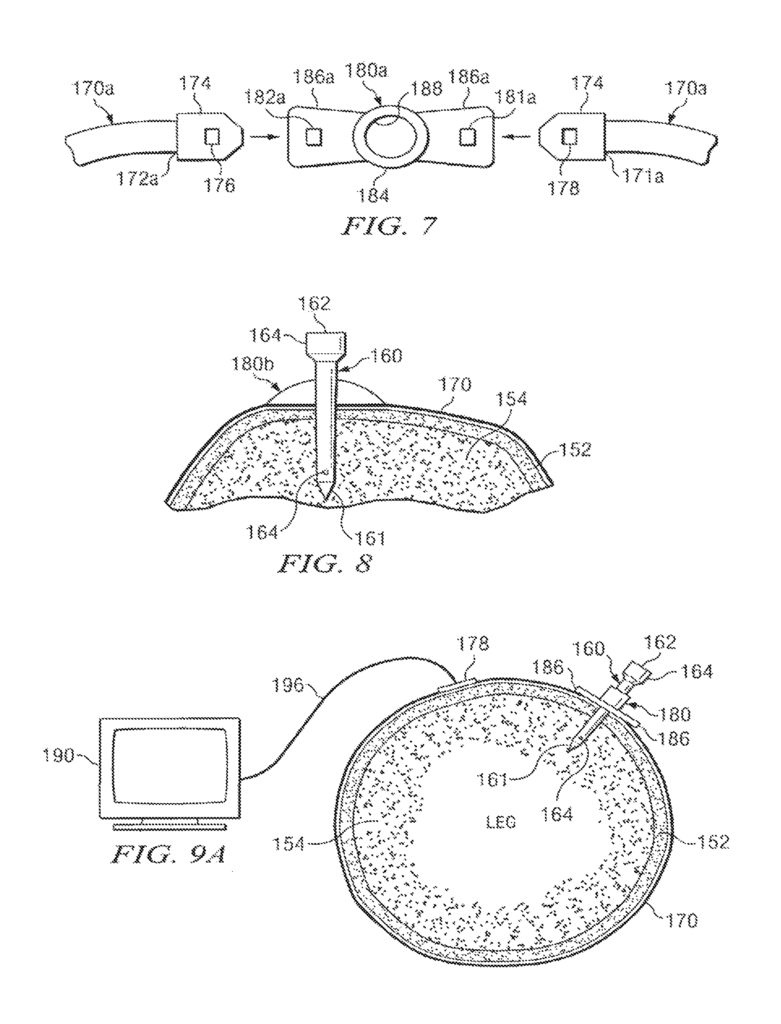
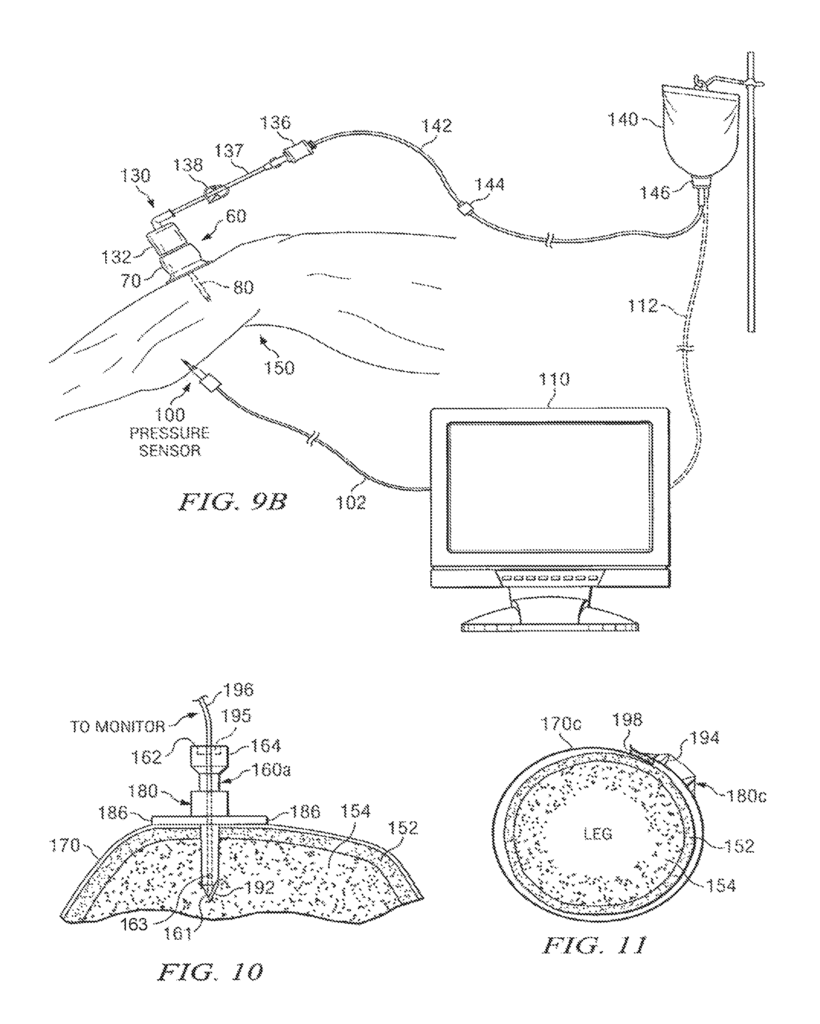
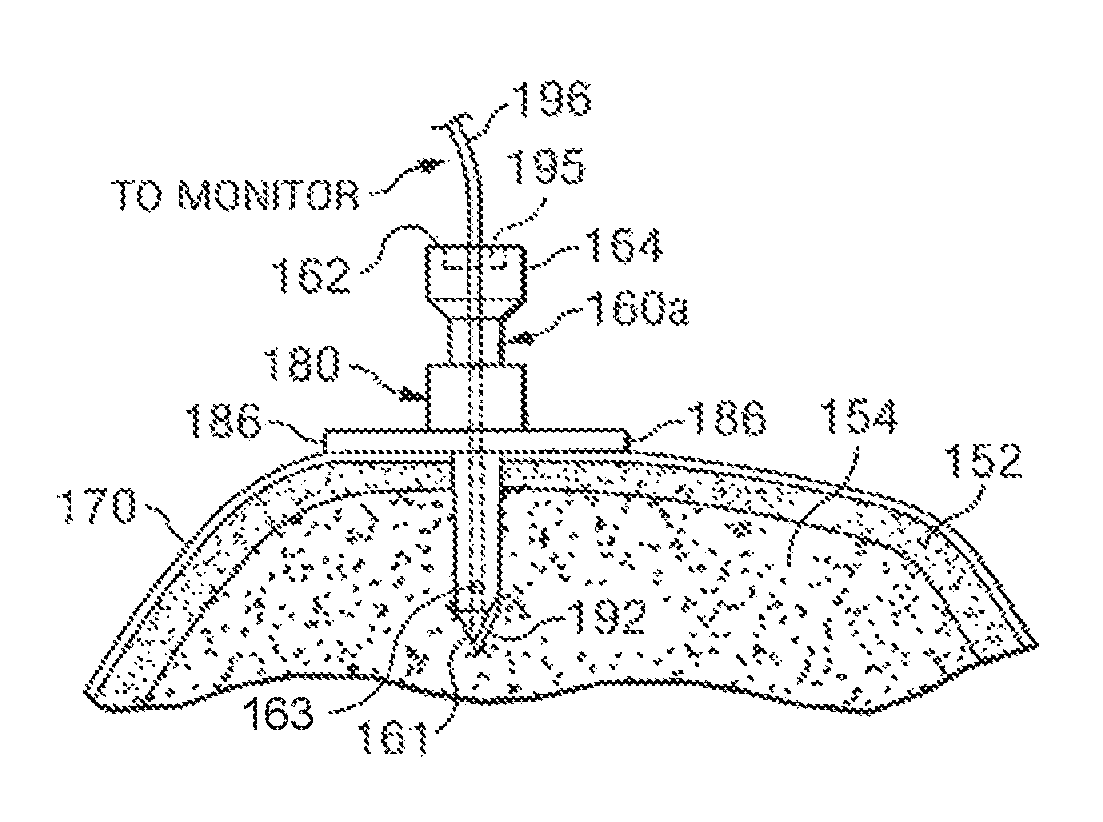
A device and method for monitoring the performance of an intraosseous devices may be available. This includes a system that comprises an intraosseous, a sensor within the tip of the intraosseous, a monitor to record the signal from it, and an electric conductor coupled with the sensor and capable of transmitting the signal from its sensor to the monitor. The tip of an intraosseous device can be designed to penetrate bone and bone-marrow so that it is placed in the bone. The tip may include a port that allows fluids to be delivered into the bone marrow.
Background for Apparatus for monitoring, supporting and/or installing intraosseous devices
Vascular accessibility is essential for the viability of a person in an emergency situation, while being transported to a medical facility or during treatment at that facility. Accessing vascular access can be a problem for five to ten per cent of patients, of all ages and sizes, in both pre-hospital and in hospital settings. This is equivalent to six (6) millions patients annually in the U.S. Patients with conditions like shock, cardiac arrest and drug overdose, as well as patients who are dehydrated, diabetic coma, kidney failure, or altered states of consciousness, may not have many (if any) veins.
Central line access can be used in a hospital or similar facility as an alternative to IV access. Central line access can be more expensive, takes longer and is more likely to cause complications. It also requires skilled personnel to insert the central lines correctly. Many hospital settings are seeing physicians and nurses increasingly opt for intraosseous (IO), rather than central line access. Paramedics and other emergency medical services (EMS) providers in pre-hospital settings often find that IO access can be fast, safe, and effective when IV placement becomes difficult.
The intraosseous space is a non-collapsible vein that can be used to infuse drugs, blood, and other fluids into patients’ central circulation. It does this within seconds and often with little discomfort. Current guidelines suggest that IO Access may be the standard of care for cardiac arrest patients. This is because IO access is comparable to central line access in efficacy, and may have fewer complications for patients and EMS providers.
In accordance to the teachings of this disclosure, apparatuses and methods are provided that allow for access to a patient?s vascular system and monitor any changes. An intraosseous (IO), device incorporating the teachings of this disclosure may be placed at certain insertion sites or target areas in order to infuse drugs and transmit fluids to a patient’s bone. To enhance the performance of different types of IO devices, support structures and attachment techniques may be included in the teachings of this disclosure.
One aspect may be to provide apparatus and methods for stabilizing, or securing, an intraosseous device that is disposed in bone or other soft tissue. A wide range of intraosseous devices can be supported using attachment devices, support structures and attachment techniques that incorporate the teachings of this disclosure.
Another aspect to the disclosure could include the use one or more sensors for monitoring performance of an intraosseous instrument during infusion of drug and/or fluid communication with a patient’s cardiovascular system.
Another aspect to the present disclosure could include a system to monitor performance of an intraosseous devices. It would consist of an intraosseous, a sensor and a monitor that records a signal from it. An electrical conductor connected to the sensor and used to transmit the signal to the monitor. An intraosseous device could include a tip that can penetrate bone and bone, with an end opposite the tip designed to be outside the bone. A longitudinal bore extends from the tip to its opposite end. The tip of an intraosseous device may contain the sensor. A pressure transducer may be used to measure pressure.
The present disclosure could provide apparatus and methods for establishing vascular access during treatment at many locations and facilities including but not limited to accident sites, battlefields and emergency medical services (EMS), facilities, oncology treatments centers, chronic disease treatment areas and veterinary applications.
Preferred embodiments and their advantages can be best understood using FIGS. 1-15B, where like numbers refers to the same and similar parts.
Access to the vasculature may be crucial for many serious conditions, chronic conditions, and emergency situations. Many patients have difficulty accessing intravenous (IV), effective treatment due to their inability to do so. The intraosseous (IO), space is a direct route to the patent’s systemic circulation and vascular system. IO access can be used to administer a variety of medications, fluids, and drugs. The rapid IO access method is a great option for any serious emergency that calls for vascular access to deliver life-saving drugs, other medications, and/or fluids in cases where traditional IV access is not possible or difficult.
The insertion site for an IO device that establishes access to a patient?s vascular system may be found in the patient’s upper tibia proximate his knee. An insertion site for IO access may be found in the humerus of a patient?s arm. Teachings of the present disclosure do not only apply to human patients. Many of the teachings from this disclosure can also be applied to animals in a veterinarian practice.
IO access can be used to?bridge? Temporary fluid and drug treatment) in emergency situations until more permanent IV sites are available. Fluids and/or medication administered via an IO Access may be used to stabilize patients and expand veins. The present disclosure could be used to teach IO devices and related procedures how to administer fluids and medications in cases where IV access is not possible or difficult.
Intraosseous Access may be used as a routine? Patients with chronic conditions that significantly reduce or eliminate access to conventional IV sites. These chronic conditions include patients on dialysis, patients with severe illnesses in intensive care units, and epilepsy. Intraosseous devices, with supporting structure or monitoring equipment incorporating the teachings of this disclosure, may be used quickly and safely to allow IO access to a patient?s vascular system in difficult situations such as status epilepticus. This will enable medical personnel to administer vital medications and fluids. Additional examples of chronic and acute conditions such as these are provided near the end.
When a patient is moving, or has the potential of moving, it may make it difficult to insert an intraosseous device (IO) at a desired insertion location. Patient safety is at risk if an IO device is inserted in the wrong spot. Patients with epilepticus and violent patients (drug overdoses, mental changes) may have difficulty moving around. This is something that must be managed for their safety. Patients with epilepsy may be agitated for long periods of time, making it difficult to start a conventional IV. It may also be difficult to place an IO device in a desired insertion location for epileptic patients.
Support structures, attachment mechanisms, and attachment techniques incorporating the teachings of this disclosure may help to minimize problems when inserting an IO device. These attachment techniques, attachment mechanisms, and supporting structures may be simple to use, even in challenging field environments.
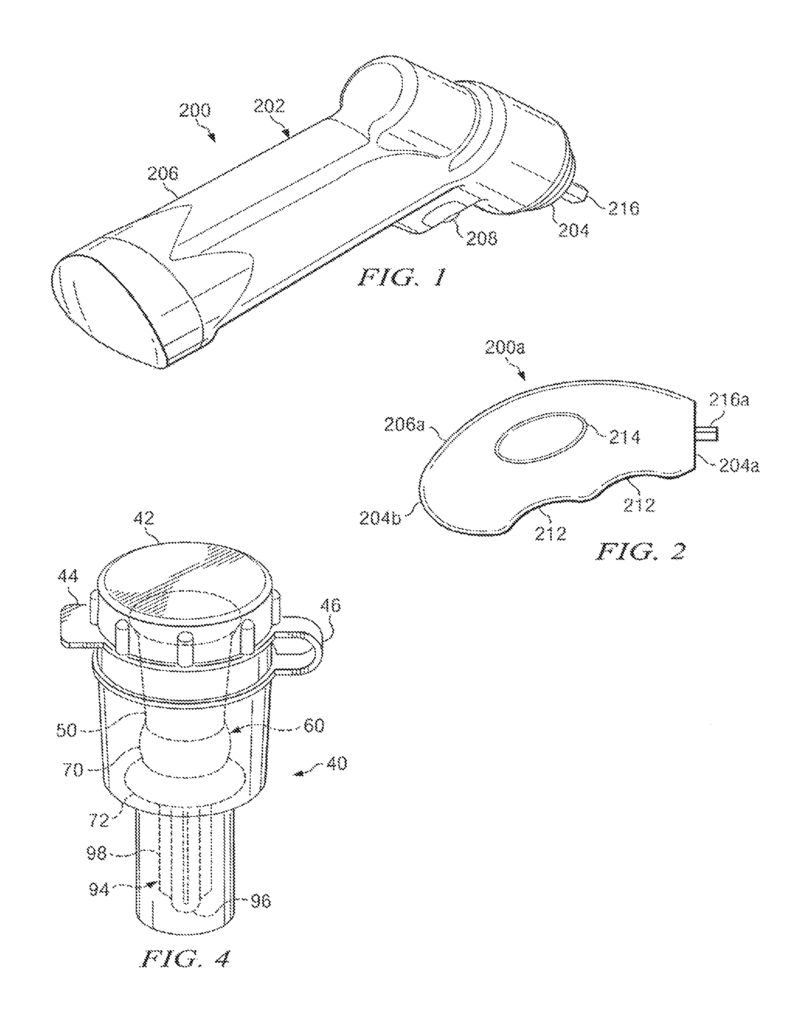
Driver” is a generic term that can be used to describe any type of driver or powered driver. “Driver” can be used to refer to any type of driver, powered or manual, that is capable of inserting intraosseous (IO), devices into selected areas of a patient’s vascular systems.
There are many techniques that can be used to releasably attach or engage an IO device, and/or penetrator assembly with manual drivers or powered drivers. A powered driver or a manually driven driver can be used to directly couple an IO device with some applications. Other applications may require the use of different types connectors to connect a manual or powered driver with an IO. You can use a wide range of connectors, connector receptacles and fittings to releasably connect an IO device using a manual or powered driver.
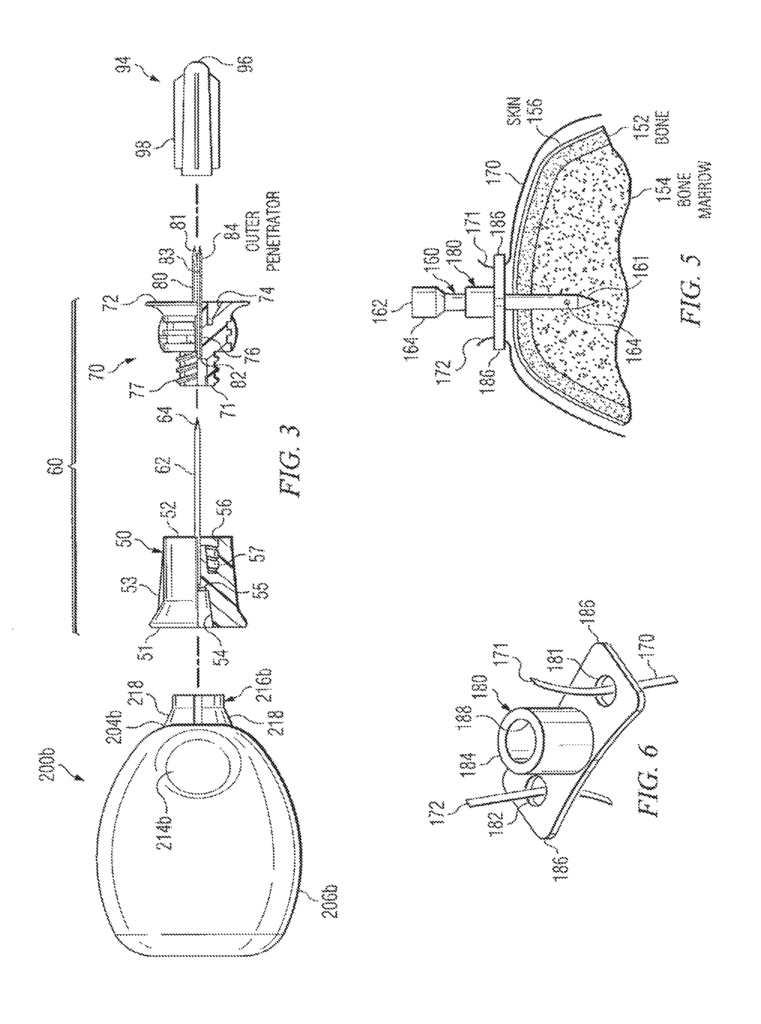
Intraosseous (IO), device” is the term used. This application may include any hollow needle, hollow drill bits, penetrator assembly or bone penetrator. During installation at a target site, a variety of trocars and spindles may be placed within the cannula. These trocars, shafts, and spindles can also be called inner penetrators. An outer penetrator could be described as a cannula.
The term “fluid” is defined as: “Fluid” may be used in this patent application to refer to any liquid, including blood, water and saline solutions. Fluid is a term that can be used to describe fluids. This patent application may also include fluids, such as blood and cells, which can be taken from a target location.
Various features may be described in relation to the powered driver 200 and/or manual driving 200 a, and 200 b. The present disclosure can also be used to describe intraosseous devices 60 160 160 160 a. Supporting structures, attachment mechanisms, and attachment techniques that incorporate teachings from the present disclosure can be used satisfactorily with many drivers and intraosseous device types. The present disclosure does not limit its use to intraosseous devices 60 or 160 a, or drivers 200, 200a, or 200b.
Powered driver 200″ may contain housing 202, which could be equipped with different types of motors or gear assemblies (not shown). Housing 202 may contain a rotatable shaft, not shown. It can be connected to a gear assembly (not shown). There are many types of fittings, connections and connectors that can be found at the shaft’s end, which extends from housing 202’s end 204.
Click here to view the patent on Google Patents.
Leave a Reply