Invented by Cameron S. Brandt, Jacob J. Kennedy, Wenfeng Xu, Eugene C. Yi, Brian A. Fox, Zeren Gao, Pallavur V. Sivakumar, Zymogenetics Inc
The B7 family of proteins plays a crucial role in regulating immune responses and has gained considerable attention in the field of immunotherapy. Among the B7 family members, zB7H6 has emerged as a promising target for cancer immunotherapy due to its unique expression pattern and ability to activate natural killer (NK) cells.
One of the key factors driving the market for zB7H6 and related compositions is the increasing prevalence of cancer worldwide. According to the World Health Organization (WHO), cancer is one of the leading causes of death globally, with approximately 9.6 million deaths in 2018. This alarming statistic has led to extensive research and development efforts to discover novel immunotherapeutic targets, such as zB7H6, to combat cancer effectively.
The potential applications of zB7H6 and related compositions extend beyond cancer immunotherapy. Recent studies have shown its involvement in autoimmune diseases, infectious diseases, and even neurological disorders. This broadens the market opportunities for zB7H6 and opens avenues for the development of innovative therapeutic strategies.
Furthermore, the advancements in biotechnology and genetic engineering techniques have facilitated the production of recombinant zB7H6 proteins and antibodies. These techniques have enabled researchers and pharmaceutical companies to explore the therapeutic potential of zB7H6 in preclinical and clinical studies. As a result, the market for zB7H6-related compositions, such as monoclonal antibodies and fusion proteins, is expected to witness substantial growth in the coming years.
In terms of market players, several biotechnology and pharmaceutical companies have recognized the potential of zB7H6 and are actively engaged in its development. Collaborations and partnerships between academic institutions, research organizations, and industry players have become common to accelerate the discovery and development of zB7H6-based therapeutics.
However, challenges such as regulatory approvals, manufacturing complexities, and high development costs may hinder the market growth to some extent. The regulatory landscape for immunotherapies is evolving, and obtaining approvals for novel targets like zB7H6 may require extensive clinical data and rigorous safety assessments. Additionally, the production of zB7H6-based therapeutics on a large scale can be challenging, leading to higher manufacturing costs.
Despite these challenges, the market for zB7H6 and related compositions and techniques holds immense potential. The increasing investments in research and development, coupled with the rising demand for effective cancer therapies, are expected to drive the market growth. Moreover, the expanding applications of zB7H6 in various disease areas provide opportunities for market expansion beyond oncology.
In conclusion, the market for the B7 family member, zB7H6, and related compositions and techniques is witnessing significant growth and potential. The increasing prevalence of cancer, advancements in biotechnology, and expanding applications of zB7H6 contribute to the market’s positive outlook. While challenges exist, collaborations and investments in research and development are expected to overcome these hurdles and pave the way for innovative zB7H6-based therapeutics in the future.
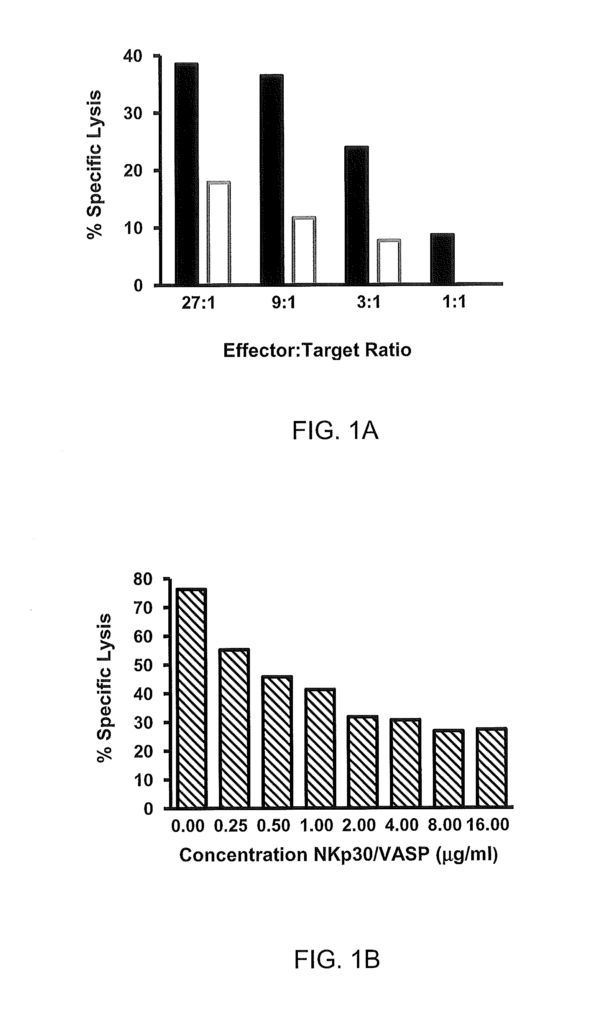
The Zymogenetics Inc invention works as follows
Disclosed are methods and compositions for modulating NKp30-mediated NK cell activity based on the interaction of zB7H6 with NKp30, as well as related screening methods. The disclosure also includes methods and compositions that modulate NKp30 mediated NK cell activation based on zB7H6’s interaction with NKp30. Related screening methods are also disclosed. Anti-zB7H6 antigens and antibody-drug combinations containing an anti-zB7H6 antigen conjugated with a therapeutic agent are also disclosed.
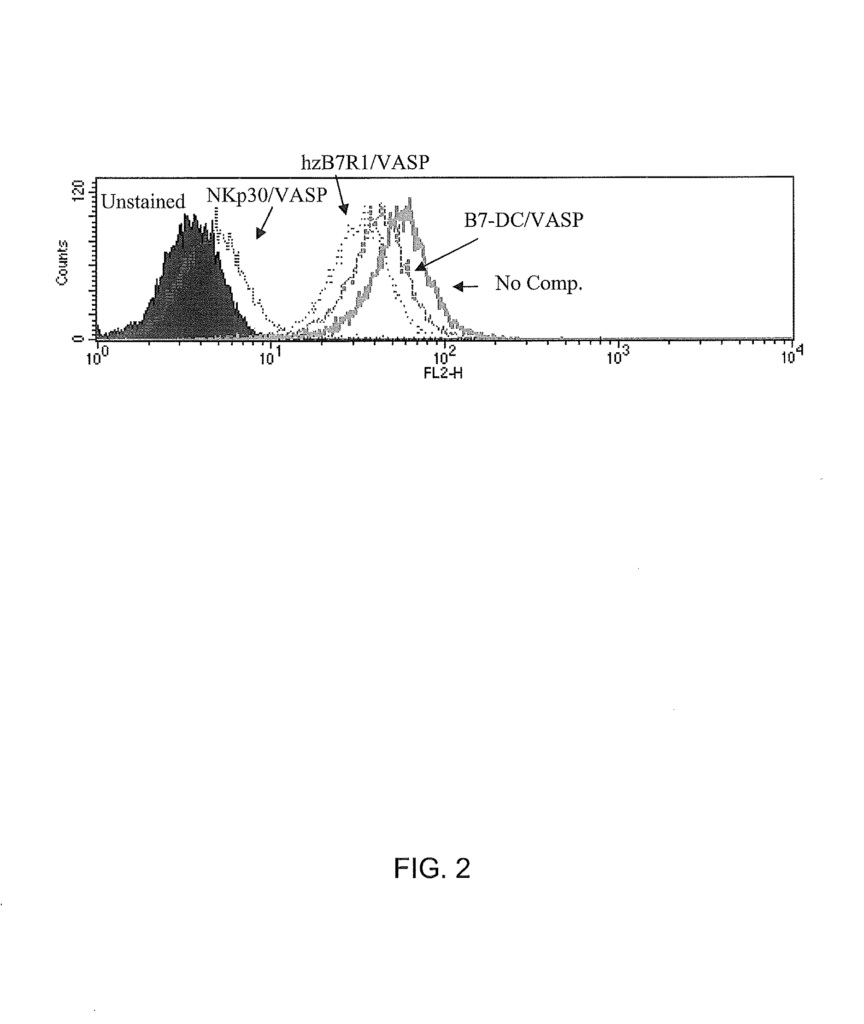
Background for The B7 family member, zB7H6, and related compositions and techniques
B7 Family
The molecules that transmit these signals are effective targets for immunomodulatory drugs. CD28, a prototypical T-cell costimulatory protein, can emit signals in response to the engagement of the T-cell receptor (TcR). The CD28 homologue, cytotoxic lymphocyte antigen-4, however, inhibits T-cell proliferation and function. (See Chambers et al., Ann. Rev. Immunol., 19:565-594, 2001; Egen et al., Nature Immunol., 3:611-618, 2002.)
Several new molecules that are homologous to the B7 families have been discovered” (Abbas and al., Nat. Med., 5:1345-6, 1999; Coyle et al., Nat. Immunol., 2: 203-9, 2001; Carreno et al., Annu. Rev. Immunol., 20: 29-53, 2002; Liang et al., Curr. Opin. Immunol. 14: 384-390, 2002) and their role in activating lymphocytes is only just beginning to become clear. These new costimulatory receptors include B7h2, PDL1, PDL2, B7H3 and B7H4.
The expression of B7 family members on antigen-presenting cell is mostly restricted. These studies collectively revealed that members of the B7 family are counter-receptors that are expressed on lymphoid cell surface receptors that interact with cognate lymphocyte receptors to produce positive or negative costimulatory signal that plays a critical role in regulating cell-mediated immunity.
Accordingly there is a requirement in the art to identify additional B7 members, their anti-receptors and molecules derived from them that have lymphocyte stimulatory activity. This is due to their biological importance, and the therapeutic potential that agents with the ability to affect their activity have. These agents that modulate costimulatory signaling would be highly useful in modulating immune responses and are highly desired.
NK Cells” and NKp30
NK cells represent an important arm in innate immunity, and they play a major role in the immune surveillance of tumors and viruses-infected cells. Unless activated, NK-cells are ineffective at performing their normal function even if they are present in sufficient numbers. In fact, decreased NK cells activity is linked to cancer and infectious disease (see Yamazaki et. al., Oncology reports 9:359-363 2002; Rosenberg et. al. Cancer Research 51:5074-5079(suppl. ), 1991; Britteenden et al., Cancer 77:1226-1243, 1996; U.S. Pat. Nos. Nos. As noted above, NK cells mediate acute rejection of BMC Allografts. “NK cell levels appear to be important in immune-related disorders.
The interaction between MHC class 1 molecules and inhibitory or activating receptors is usually what regulates NK cell activity. (See, e.g. Barao and Murphy BB & MT 9, 727-741,2003.) The “missing self” The original hypothesis was based on the observation of tumor cells lacking MHC class 1 molecules being susceptible to NK cell killing. (See Ljunggren & Karre, Immunol. Today 11:237-244, 1990; Ohlen et al., J. Immunol. 145:52-58, 1990.) Investigators additionally observed that human NK cells lyse class-I-deficient Epstein-Barr-virus-transformed B-lymphoblastoid cell lines. (Storkus et al., Proc. Natl. Acad. Sci. USA 86:2361-2464, 1989. It was also found that the transfection of class-I genes into class-I-deficient cells made these cells partially or completely resistant against NK-cell-mediated lysis. (See Storkus and DeMars in Eur. J. Immunol., 19:447-451, 1989.) MHC class 1 is not necessary to protect against NK-cell mediated cytotoxicity. Recognition by MHC Class I does not prevent cytolysis of NK cells. (Barao & Murphy, supra.). During recent years, various MHC-class-I-specific inhibitory and activating receptors as well as non-MHC-class-I-specific activating receptors have been identified. These receptors have relevance for therapeutic approaches, such as allogeneic BMT or cancer therapy. (See id.)
Non-MHC-class-I-specific activating receptors, which are capable of mediating NK cell cytotoxicity against MHC-class-I-deficient or negative targets, are represented in part by a heterogeneous family of NK cell-specific immunoglobulin-like molecules that are known as natural cytotoxicity receptors (NCRs). (See, e.g., Moretta et al., Annu. Rev. Immunol. Immunol. Rev., 181:170-184, 2001.) These activating receptors are activated by ligation on NK-cells in the absence of MHC-class I expression, such as on virus-infected or tumor-cells. One of these activating receptors is NKp30. It is expressed constitutively and selectively on mature natural killer cells (NK). This receptor signals, among other things, through coupling with CD3*. (See Barao & Murphy, supra.). “The target-cell ligand that NKp30 binds to has not previously been identified.
This system of innate recognition of NK cells is a powerful tool that could be used in clinical applications such as allogeneic bone marrow (BMT) transplantation, cancer therapy or the treatment of other NK cell-associated disorders. (See e.g. Barao & Murphy, supra.). In order to modulate NK cell function and treat diseases or disorders that are associated with NK cells, it would be beneficial to stimulate or inhibit activation of NKp30. The activation of NKp30 is useful in treating diseases and disorders characterized by low NK activity, including cancer, infectious disease, and other disorders. Inhibition of NK activity by blocking NKp30, on the other hand, would be helpful for treating NK-cell mediated disorders such as BMC allograft reject. “Those skilled in the art will be able to discern from this disclosure that the present invention includes compositions and methods suitable for treating these diseases and disorders.
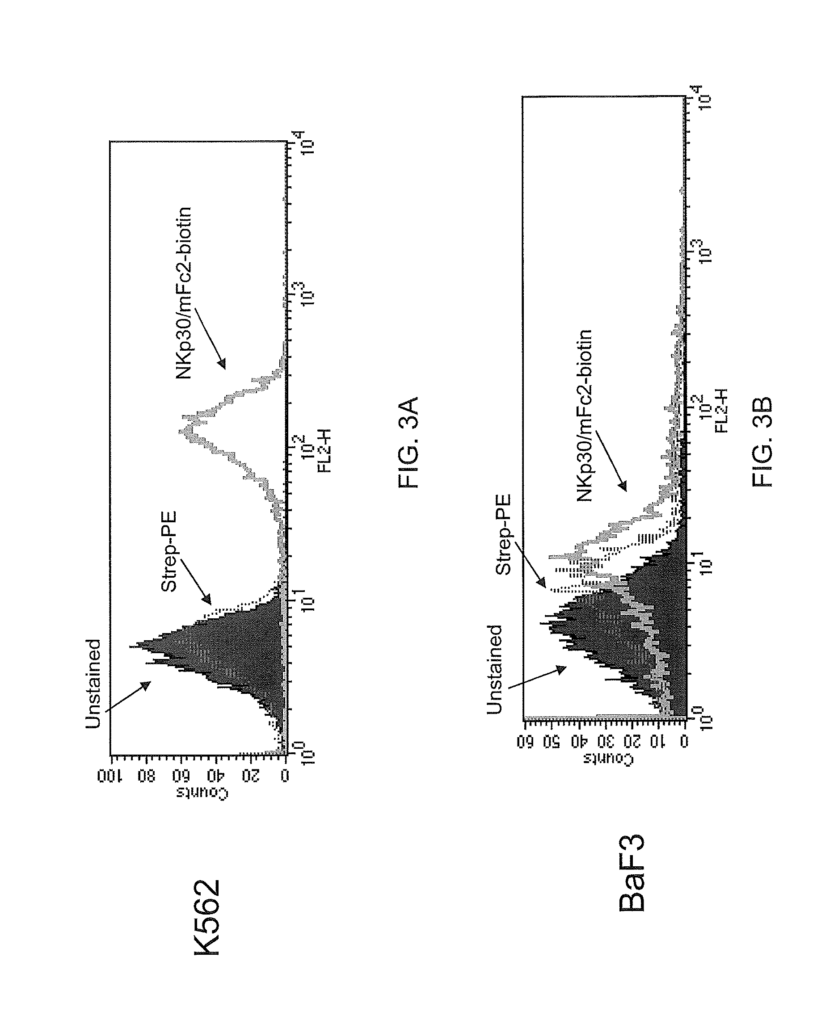
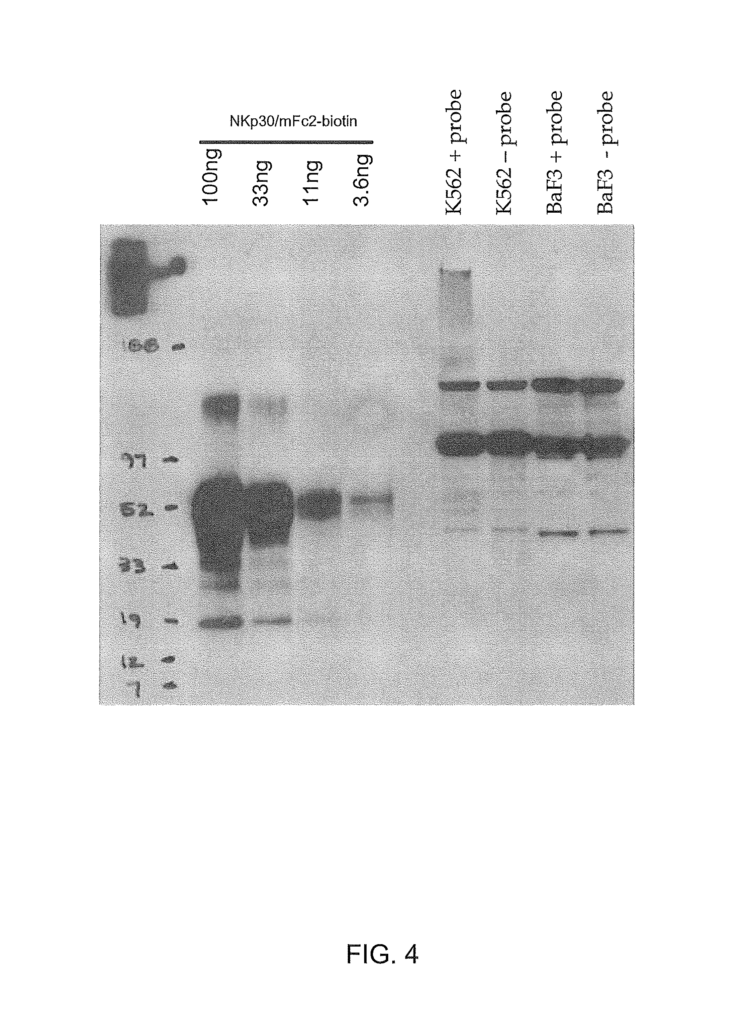
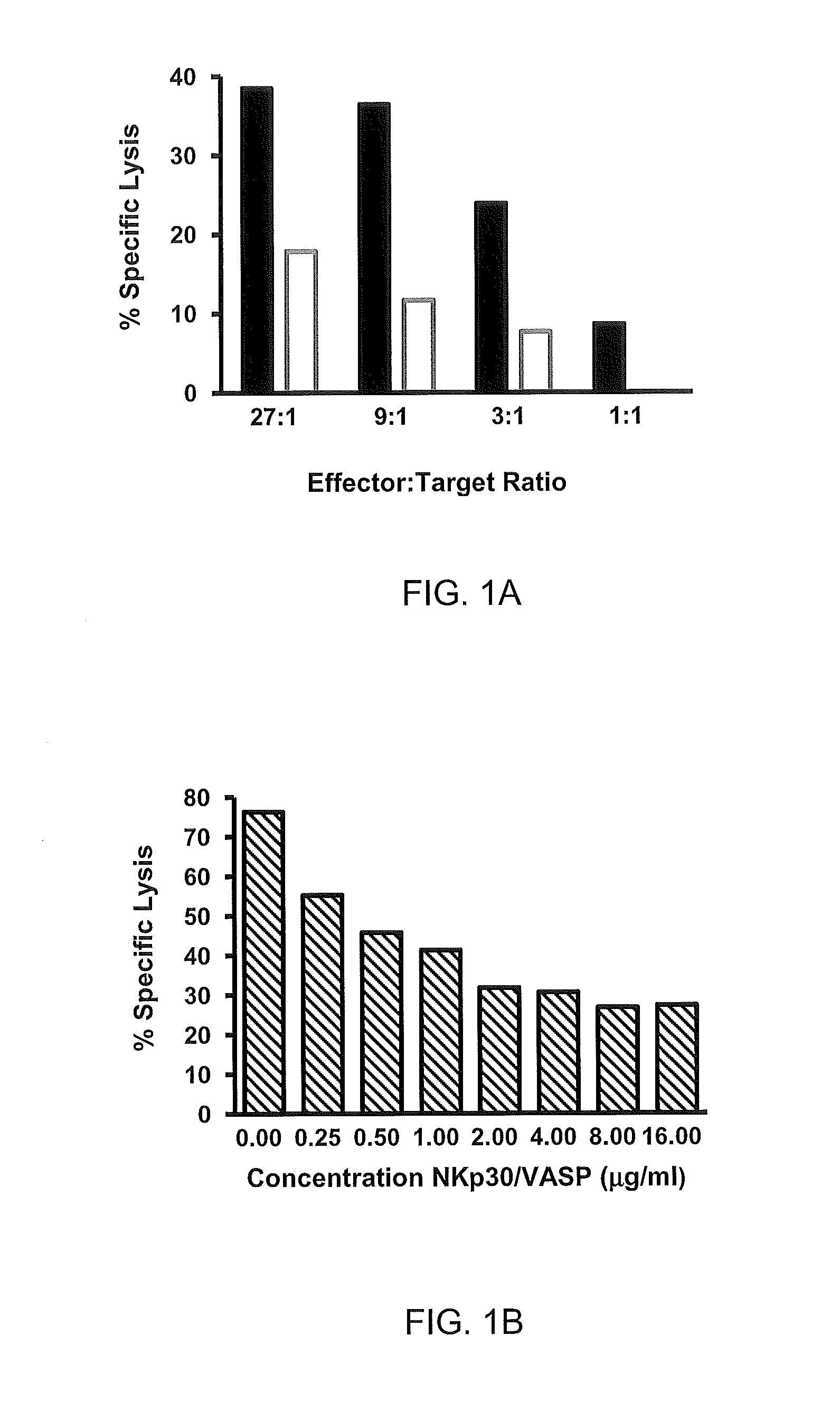
The present invention, in one aspect, provides isolated zB7H6 peptides including polypeptide-fusions that contain the amino acid sequence from SEQ ID No:2 or functional variants or fragments thereof. In some embodiments, the zB7H6 is a soluble polypeptide that is isolated and has a polypeptide sequence with at least 90% or 95% sequence similarity to the amino acid sequence of residues 25-266 in SEQ ID No:2, the soluble polypeptide being capable of binding specifically to human NKp30. In some variations, a soluble polypeptide of the invention comprises a segment polypeptide with an amino acid sequence corresponding to residues 25-266 (or 1-266) in SEQ ID No:2. Solvable fusion proteins can be an example of such soluble polypeptides. Polypeptides that are suitable soluble fusions include those polypeptides which further comprise an immunoglobulin constant heavy chain region (e.g. an Fc fragment), for example an IgG, IgM (e.g. IgG1, IgG2, IgG3, IgG4, or IgG4) or IgE (e.g. IgG1, IgG2, IgG3, IgG4, or IgG4). Polypeptides that contain a VASP-domain are also suitable soluble proteins.
In another aspect, this invention provides isolated polynucleotides that encode a zB7H6 peptide. In certain embodiments the present invention provides a isolated polynucleotide that comprises a segment of polynucleotide encoding a solubilized zB7H6 peptide. The zB7H6 peptide includes a segment of polypeptide having at least 90% sequence similarity with the amino acids sequence in residues 25-266 in SEQ ID No:2, wherein the polypeptide can specifically bind to human NKp30. In one variation, the encoded polypeptide soluble zB7H6 comprises a segment of polypeptide having the amino acids sequences set forth in SEQ ID No:2. The encoded polypeptide could be a soluble protein such as an immunoglobulin constant region or VASP domain. In some variations, the polynucleotide sequence encoding the polypeptide zB7H6 comprises nucleotides 1-798 or 73-798 from SEQ ID No:1.
In yet another aspect, the present invention also provides vectors including expression vectors that include a polynucleotide in the same manner as described above. In some embodiments of the invention, an expression vector comprises the following operably-linked elements: a transcription initiating region; a segment of DNA encoding a soluble polypeptide; the polypeptide segment of the zB7H6 contains a sequence that is at least 90% identical to the amino acid sequence of residues 25-266 in SEQ ID No:2, and the polypeptide can bind specifically to human NKp30. The present invention also provides host cells containing such vectors as well as methods to produce a polypeptide zB7H6. In certain embodiments, the method for producing a soluble polypeptide zB7H6 includes cultivating a host cell containing an expression vector, as described above, under conditions where the polypeptide will be expressed, and recovering that expressed polypeptide.
The present invention provides isolated antibodies which specifically bind to the zB7H6 peptide described herein.” In certain embodiments of the invention, an antibody is provided that binds specifically to a polypeptide with the amino acid sequence described in residues 25-266 in SEQ ID No:2. In certain variations, the antibodies inhibit the interaction between zB7H6 and human NKp30. Monoclonal antibody, such as humanized or human monoclonal antigens, are particularly suitable. Single chain antibodies are also included in the anti-zB7H6 antibody family.
The present invention also provides methods to modulate the activity of human natural killer cells (NK). One method involves enhancing NK cells activity by contacting a NK cell with a recombinant membrane-bound zB7H6 peptide. The zB7H6 peptide comprises a polypeptide with at least 90% sequence similarity to the amino acid residues 25-266 in SEQ ID No:2, where the zB7H6 peptide can specifically bind to human NKp30. In one variation, the zB7H6 segment contains the amino acid sequence of residues 25-266 in SEQ ID No:2.
Other methods of modulating NK cells activity include decreasing NK cell activities against a zB7H6 expressing cell. These methods involve contacting a zB7H6-expressing cell in the presence a human NKT cell with a sufficient amount of an antigen that binds specifically to the polypeptide segment with the amino acid sequence of residues 25-266 in SEQ ID No:2, and where the antibody inhibits zB7H6’s interaction with human NKp30. These methods of decreasing NK cells activity can be used, for instance, to treat bone marrow allograft reject. In certain variations, a technique of the invention comprises treating bone marrow cells (BMC) rejection in a subject by administering, to the subject in a quantity effective to inhibit NK activity and thereby to treat acute BMC allograft reject, an antibody which (a) binds specifically to a segment of polypeptide having an amino acid sequence as set forth at residues 25-266 in SEQ ID No:2 and (b), inhibits the interaction between zB7H6 and human Monoclonal antibody (e.g. humanized or human monoclonal antibody) are particularly suitable antibodies. Single chain antibodies can be used to treat BMC.
In another aspect, the present invention provides methods for inducing antibody dependent cellular cytotoxicity (ADCC) against a zB7H6-expressing cell. Such methods generally include contacting the zB7H6-expressing cell with an effective amount an antibody that specifically binds to a polypeptide segment having the amino acid sequence set forth in residues 25-266 of SEQ ID NO:2, wherein the contacting is in the presence of an NK cell or a CD8+ T cell expressing an Fc receptor having ADCC activity, and wherein the antibody comprises an Fc region capable of binding the Fc receptor. Suitable anti-zB7H6 antibodies include monoclonal antibodies, including, for example, human or humanized monoclonal antibodies, as well as single chain antibodies. In certain variations, the Fc region is a single chain Fc (scFc). The zB7H6-expressing cell can be, for example, a zB7H6-expressing cancer cell. zB7H6 cancer cells particularly amenable to targeted killing using these methods include, e.g., colon cancer cells, liver cancer cells, cervical cancer cells, lung cancer cells, pancreatic cancer cells, prostate cancer cells, prohemocytic leukemia cells, B-cell lymphoma cells, monocytic lymphoma cells, erythroleukemia cells, Burkitt’s lymphoma cells, and chronic myelogenous leukemia cells.
In a second aspect, the invention provides methods to induce complement-dependent cytotoxicity against a zB7H6 expressing cell. These methods include contacting a zB7H6 expressing cell in the presence complement with a sufficient amount of an anti-zB7H6 antibodies that binds specifically to a polypeptide sequence corresponding to residues 25-266 in SEQ ID No:2. Anti-zB7H6 monoclonal antibody, such as humanized monoclonal or human monoclonal, and single chain antibodies are suitable. In some variations, the Fc is a single-chain Fc. A zB7H6 expressing cell could be a cancer cell, for instance. “ZB7H6 cancers cells are particularly susceptible to targeted killing with these methods, including colon cancer, liver cancer, cervical cancer, lung cancer, pancreatic, prostate, B-cell, lymphoma, monocytic, and erythroleukemia, as well as Burkitt lymphoma, erythroleukemia, and chronic myelogenous lymphoma.
The present invention also provides methods to treat a cancer that expresses zB7H6 in a patient. These methods include giving the subject a sufficient amount of an anti-zB7H6 antibody which binds specifically to a polypeptide having an amino acid sequence of residues 25-266 in SEQ ID No:2, and where the antibody has an Fc region with ADCC or CDC activity. Anti-zB7H6 monoclonal antibody types include humanized monoclonal and human monoclonal, as well single chain antibodies. In some variations, the Fc is a single-chain Fc. Cancers that express zB7H6 are particularly susceptible to being treated using these methods. These include cancers in the colon, liver and cervix as well as pancreas, lungs, prostate, erythroleukemia or Burkitt’s leukemia.
The present invention also provides an antibody-drug combination that contains an antibody which binds specifically to a polypeptide having an amino acid sequence as set forth by residues 25-266 in SEQ ID No:2, and where the antibody is coupled to a toxic agent. In some embodiments, an antibody that binds to the amino acid residues 25-266 in SEQ ID No:2 is monoclonal, such as a humanized or human monoclonal. In some variations, the monoclonal antibody is used. Anti-tubulins, DNA minor grove binding agents, DNA Minor Groove Alkylating Agents, Duocarmycins and Puromycins are all suitable cytotoxic agents. Anti-tubulin agents that are particularly suitable include dolastatins (vinca alkaloids), podophyllatoxins (podophyllatoxins), taxanes (taxanes), baccatin derivatives and cryptophysins.
Click here to view the patent on Google Patents.
Leave a Reply